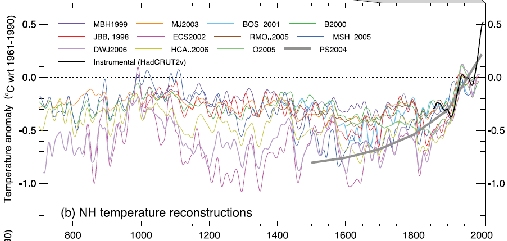
Much research effort over the past years has gone into reconstructing the temperature history of the last millennium and beyond. The new IPCC report compiles a dozen reconstructions for the temperature of the Northern Hemisphere (including of course the original “hockey stick” reconstruction, despite opposite claims by the Wall Street Journal). Lack of data does not permit robust reconstructions for the Southern Hemisphere. Without exception, the reconstructions show that Northern Hemisphere temperatures are now higher than at any time during the past 1,000 years (Figure 1), confirming and strengthening the conclusions drawn in the previous IPCC report of 2001.
Fig. 1: Figure 6.10 (panel b) from the paleoclimate chapter of the current IPCC report (see there for details).
“Climate sceptics” do not like this and keep coming up with their own temperature histories. One of the weirdest has been circulated for years by German high-school teacher E.G. Beck (notorious for his equally weird CO2 curve). This history shows a medieval warm phase that is warmer than current climate by more than 1 ºC (see Figure 2). So how did Beck get this curve?
Fig. 2, modified from E.G. Beck (we added the green parts).
The curve is a fake in several respects. It originally is taken from the first IPCC report of 1990: a scan of the original is shown in Figure 3. At that time, no large-scale temperature reconstructions were available yet. To give an indication of past climate variability, the report showed Lamb’s Central England estimate. (Unfortunately this was not stated in the report – an oversight which shows that IPCC review procedures in the early days were not what they are now. We will post in more detail on the history of this curve another time.)
Fig. 3. The past millennium as shown in the first IPCC report of 1990, before quantitative large-scale reconstructions were available. This curve was based on Lamb’s estimated climate history for central England.
But Beck did not stop at simply using this outdated curve, he modified it as highlighted in green in Figure 2. First, he added a wrong temperature scale – the tick marks in the old IPCC report represent 1 ºC, so Beck’s claimed range of 5 ºC exaggerates the past temperature variations by more than a factor of three. Second, the original curve only goes up to the 1970’s. Since then, Northern Hemisphere temperatures have increased by about 0.6 ºC and those in central England even more – so whatever you take this curve for, if it were continued to present, the current temperature would be above the Medieval level, as in the proper reconstructions available today. As this would destroy his message, Beck applied another fakery: he extended the curve flat up to the year 2000, thereby denying the measured warming since the 1970s. With this trick, his curve looks as if it was warmer in Medieval times than now.
When approached directly about these issues, Beck published a modified curve on a website. He changed the temperature range from 5 ºC to 4.5 ºC – but he shortened the arrow as well, so this was just cosmetics. He also added instrumental temperatures for the 20th Century at the end – but with his wrong temperature scale, they are completely out of proportion. (In fact his version suggests temperatures have warmed by 2 ºC since 1900, more than twice of what is actually observed!)
Beck goes even further: in a recent article (in German), he has the audacity to claim that his manipulated curve is right and the more recent scientific results shown by IPCC are wrong. And for years, he has offered his curve on an internet site (biokurs.de) that distributes teaching materials for schools, with support from German school authorities. It is quite likely that his fake curve has been shown (and will continue to be shown) to many school children.

Re#350, DM – where have you been looking?
See also Global Surface Air Temperature in 1995: Return to Pre-Pinatubo Level, Hansen et al 1995 (pdf)
https://www.realclimate.org/index.php/archives/2006/05/current-volcanic-activity-and-climate/
http://www.sciencemag.org/cgi/content/abstract/296/5568/727
Global Cooling After the Eruption of Mount Pinatubo: A Test of Climate Feedback by Water Vapor (Science, 2002)
Brian J. Soden, Richard T. Wetherald, Georgiy L. Stenchikov, Alan Robock
“The sensitivity of Earth’s climate to an external radiative forcing depends critically on the response of water vapor. We use the global cooling and drying of the atmosphere that was observed after the eruption of Mount Pinatubo to test model predictions of the climate feedback from water vapor. Here, we first highlight the success of the model in reproducing the observed drying after the volcanic eruption. Then, by comparing model simulations with and without water vapor feedback, we demonstrate the importance of the atmospheric drying in amplifying the temperature change and show that, without the strong positive feedback from water vapor, the model is unable to reproduce the observed cooling. These results provide quantitative evidence of the reliability of water vapor feedback in current climate models, which is crucial to their use for global warming projections.”
A cynical biochemist who doesn’t know how to use Google Scholar? That’s a first.
Barton Paul Levenson (#348) wrote:
I am not complaining about what you have posted, but good god, I can’t wait until there is a more widely available, well-formatted display of mathematical notation on the web – particularly something which is user-friendly. Just displaying an integral or differential equation and some greek letters would be a real pain on blogs – and then it would look quite amateurish, assuming the symbols didn’t come out looking like complete gibberish. One line equations feel like a straight jacket, and if they were to express anything high-level would be a real pain to read. A person ought to be able to focus on what the equations are representing, not the formatting or poorly-formatted way in which they are displayed.
For those wanting specifc info regarding the spectrum of CO2, H2O etc. see.
http://www.spectralcalc.com/spectralcalc.php
For all those wanting specific spectral info on CO2, H2O etc…
http://www.spectralcalc.com/spectralcalc.php
Re: #352 (Timothy Chase)
I agree that displaying mathematics on the web can be problematic. For my own blog this is not a problem, because it’s hosted by wordpress, which allows latex (hooray for wordpress!).
One solution on your own webpage, if you have access to a latex processor, is to do the equations in latex, produce a pdf (or dvi) version to display on your screen, do a screen capture, then save an image of the equation.
But of course forums generally don’t allow the display of image files. So if I really needed to refer to equations in a comment here, I’d probably link to my blog. Another possibility is to insert (in a plain text comment like this), the tex source (e.g., P(x) = e^{-frac{1}{2} x^2 / sigma^2}). Of course that only helps the readers who comprehend tex.
[Response: You can use latexrender here. i.e. enclose the tex with [ tex ] and [ / tex ] (spaces removed) to get: . – gavin]
. – gavin]
Hansen
“Global surface air temperature has increased about 0.5°C from the minimum of mid-1992, a year after the Mt.Pinatubo eruption.”
DocMartyn
“Global surface air temperature has increased about 0.5°C from the minimum of mid-1984, a year after nothing happened at all.”
The first is in the abstract, and the statement is true, but at the same time is bollocks. From the data stream I linked it is clear that the temperature change post June91 is with in the general noise of the system, over a 25 year period (1980-2005). To claim anything else is not true. The temperature change in 92 and 93 are not large in the quarter century time scale. You can not take a pair of points and claim thet the difference is valid if both points are within the normal bounds of the system you are studying. June92 to June93 differed with the slope of 1980-1991/1994-2005 by 0.22 C, whereas 1984 did so by 0.2. If you have noise of 0.2 from the mean slope, then a signal of 0.22 cannot be statistically valid for a pertubence of the system. There is no 0.5C change in the global temperature, none at all, you cannot compare two actual measurements to each other, you have to compare them to the mean.
[Response: Look at figure S2 in this recent paper. http://pubs.giss.nasa.gov/abstracts/inpress/Hansen_etal_1.html – gavin]
Re gavin’s inline to DocMartyn’s (#356)
Heck, I would begin by looking at all the diagrams at the end, then reading the paper.
This is a perfect example of a given conclusion (or in this case, model) being justified by multiple independent lines of investigation where the degree to which the justification for the conclusion is far greater than the justification which it receives from any one line of investigation in isolation.
In this case, each line of investigation regarding one physical dimension of the system or another (e.g., irradiance, ppm of a given gas, temperature anomaly) consists of a great many “points” or measurements in a temporal series – and are in many cases undoubtedly grand averages of a far larger set of data, such that the uncertainty regarding each average (point in the temporal series) is far less than the uncertainty regarding any individual measurement. The fit is really impressive.
So you agree that the temperature drop was in the order of 0.2 degrees, and within the noise of the data series. It would have been nice for Hansen et al (in press) to have included the residuals. It is clear that the model is quite poor, the estimate of delta T is out by a factor of two.
Gavin, do you agree with my observation that the effect of Mt.Pinatubo eruption on the actual temperature of the Earth is approximately half that of the model presented in the manuscript.
———————————————————————
In figure 1B of the main manuscript I don’t understand the CO2 and forcing peaks in the WWII period. You do know that the British And American airforces burnt cities to the ground in this period, that the Soviets and then the Germans burnt cities in Eastern Europe and in Russia to the ground, that the British conscripted civilians to mine coal to fuel the war effor. You have CO2 going down, how on Earth do you have CO2 going down during WWII? Particulates in the Atmosphere I could just about live with, but a drop in CO2?
re: DocMartyn #356
“Global surface air temperature has increased about 0.5°C from the minimum of mid-1984, a year after nothing happened at all.”
Your statement may be true, but a rational skeptic (not denialist) would *ask*:
“A major volcano El Chichon erupted in 1982. Could its effects persist into 1984? are there other factors, like other volcanoes, or timing of El Ninos, or stratospheric conditions or differences in eruptions that would make cooling effects spikey or more persistent?”
My previous post seems to have been lost, so I will re-post it. I am trying to understand some basic principles of the greenhouse effect.
A greenhouse gas is sensitive to certain wavelengths of light, similar to how a piano string will ring when it receives sound at the pitch to which it is tuned. The gas “rings” by the bending and stretching of its molecular bonds, as shown by Archer in his figure 4-1. This molecule abosrbs a fixed amount of energy to enter this state. If undisturbed, the molecule will then re-radiate its energy at the same frequency and intensity at which it was received. So what happens to the wave if it has more energy than what the molecule needs? Does absorption not take place, or will the wave continue its travel with its intensity reduced by the amount of energy that it lost to the molecule.
The greenhouse gas molecule can also lose its vibrational energy by colliding with another molecule. This will cause the atmosphere to warm. But Ray in #334 suggests that a collision can also return a GHG molecule to its excited state, which can then radiate the energy. Does this process work equally in both directions? If so, then there is no net heat transfer, which is what Ray seems to be saying.
The “blanket model” I proposed is based on greenhouse gas energy being transferred to the surrounding air, which I believe is different from downwelling infrared radiation from the greenhouse gas warming the ground – the “reflector model”. My question remains: how much, if any, energy is directly transferred to the surrouding air by a GHG absorbing radiation?
The “radiation balance” model comes from Raymond Pierrehumbert (“raypierre”) in the RealClimate article Busy Week for Water Vapor, where he says:
Does the Stefan-Boltzmann law apply to a greenhouse gas, which is not a black body? Exactly how does temperature affect the behavior of a greenhouse gas? Is raypierre saying the only greenhouse gases that affects the radiation balance are those mainly at the top of the troposphere that radiate into space? This is very different than the blanket or reflector models where the greenhouse effect occurs lower in the atmosphere where carbon dioxide levels are lower.
Correction to #357
The second sentence shoulda read like,
“This is a perfect example of a given conclusion (or in this case, model) being justified by multiple independent lines of investigation where
the degree to whichthe justification for the conclusion is far greater than the justification which it receives from any one line of investigation in isolation.”Don’t preview when you enter…
(1)
(2)
latex notation simple doesn’t work for me. How do you do it? You also seem to need to to get one, and one just disapearse.
It seems to work now but not in the preview.
Seems to be necessary with 2 \ and one disapear when you preview
I don’t have the background to analyize this argument in a discussion I’m involved in:
There are 5 trillion metric tons of atmosphere around the earth Citation:(http://en.wikipedia.org/wiki/Earth's_atmosphere)
So 24.1 x10^6 divided by 5×10^15 = 4.82 x 10^-9 or .00482ppm CO2 contribution per year. That’s assuming none of the man made CO2 gets absorbed. So in 207 years, the total man made CO2 change in the atmosphere will be 1 part per million. Even at 10 times the man made rate, the CO2 conc would only change by 1 ppm in 20.7 years. I’m sure natural phenomena during that time will make man made CO2 contribution look like noise.
Total CO2 in the atmosphere is .038% =380ppm Citation: http://en.wikipedia.org/wiki/Earth's_atmosphere
So if man puts out 10 times the amount of CO2 than he does today after 20 years that 380ppm MIGHT go to 381 ppm.
Any Comments?
Re #352 — Tim, I know exactly what you mean. It’s a real pain to display an equation in HTML.
For what it’s worth, there are some tricks available. You can superscript something by surrounding it with < sup > and < / sup > HTML tags (with the spaces removed, of course), and subscript them with < sub > and < / sub >. (You can’t post angle brackets in HTML, to put these up I needed ampersand-lt-semicolon and ampersand-gt-semicolon.) You can also play with font sizes and fonts a bit. But it’s a tremendous pain. There really ought to be an easier way.
[[So if man puts out 10 times the amount of CO2 than he does today after 20 years that 380ppm MIGHT go to 381 ppm.
Any Comments? ]]
Yes. Your figure for annual CO2 added artificially is too low by several orders of magnitude. The actual figure is about 7 x 1012 kilograms per year.
I should point out re the above that about half the human technology CO2 is being absorbed by natural sinks (mostly the ocean) for the moment.
Re: #360 (rhkennerly)
The 5×10^15 part is correct, but the 24.1×10^6 part is not. Make that 24.1×10^9 (yes, one thousand times bigger). I suspect someone was reading a table giving CO2 emissions in thousands of metric tonnes, and thought it was simply metric tonnes.
Then the calculation gives 4.82 ppm CO2 per year.
But this is not actually correct, because “ppm” estimates the number of molecules, not their mass. CO2 is heavier than the average air molecule (CO2 atomic weight 44 amu, average air molecule about 28 amu), so the actual calculation (using these numbers) would give about 3 ppm CO2 per year. Of course, that’s a very rough calculation, since the atmospheric mass (5×10^15 tonne) is only given to one significant digit.
But you get the idea: whoever posted this comment on your discussion was off by a factor of 1000.
Re: #366 (BPL)
The 7×10^12 kg/yr (7×10^9 tonne) counts only the carbon in CO2. The CO2 itself is more like 24×10^9 tonne (24×10^12 kg).
Comments…
Just one
The figure from the Wikipedia table (List of countries by carbon dioxide emission) gives
24,126,416 (in units of 1000’s of metric tonnes) ==> 24 x10^9 tonnes !
Always check the units !
Blair, You’ve got it about half right. First, what kind of background do you have in thermo and/or stat mech? Think of it this way. A molecule can have lots of different types of energy–kinetic, electromagnetic potential, elastic potential… It works best if you think of a large number of gas molecules. The energy distribution of these molecules tends to follow the Boltzmann equation–exp(-E/kt), so although most molecules are at low energy, you’ve got a finite probability of having very high-energy molecules. There is a theorem–the equipartition theorem–that says that in equilibrium, all the different kinds of energy will tend to equalize–You’ll have about as much kinetic as electromagnetic, as vibrational as rotational… Intuitively, you can see how this would work. If you’ve got more kinetic energy, collisions will tend to excite more vibrational, rotational, etc. OK, that’s equilibrium. Now introduce a new energy source–IR photons that tend to put more energy into the vibrational states of a particular molecule. Over time, this energy will equilibrate and move into the other modes. Eventually, we reach a new equilibrium not only of the gas molecules with each other, but of gas molecules with the radiation field. Moreover, each time a photon is re-radiated, it’s 50-50 whether it goes up or down, so you get multiple shots at keeping the energy in the atmosphere or reradiating it back to ground. So, there is a net transfer of energy to the atmosphere (and so the ground) because the atmosphere has to come into equilibrium with the radiation field. Does this help?
Is raypierre saying the only greenhouse gases that affects the radiation balance are those mainly at the top of the troposphere that radiate into space?
Blair, I believe the answer is ‘yes.’ This is why denialist arguments that water vapor swamps CO2 as a greenhouse gas are so wrong. The atmosphere is very dry at the elevation where the earth actually loses heat into space.
re 364,366,368: My math and sources say 5000T tonnes or 5×10^15 Kg = 5×10^9 million Kgs of atmosphere. At roughly 380ppmv = 570ppmm (“m” for mass) = 2.85×10^12 Kg of CO2 residing in the atmosphere. My sources say the worldwide release of CO2 (2006) is about 32B tons = 29B tonnes = 2.9×10^13 Kgs. Unabsorbed this should raise the concentration by about 5.8ppkm = 0.0058ppmm = 0.0038ppmv. Is this possibly correct?? Between tons and tonnes and ppmm and ppmv my haed is spinning with this advanced math.
Mark (373) says “…denialist arguments that water vapor swamps CO2 as a greenhouse gas are so wrong. The atmosphere is very dry at the elevation where the earth actually loses heat into space”
I fail to see the connection. Water vapor, when it’s around, absorbs much more energy from the IR radiation than anything else. It tends to be a much less concentration at the top and therefore does not re-radiate as much into space. Why does this counter the “denialist’s” argument that you cite????
[Response: The key point, which we have discussed previously on this site at length, is that water vapor acts (w/a few minor exceptions, e.g. human irrigation and land use change, etc) as a feedback, not a forcing< /a>. -mike]
When Mark Zimmerman states in #373,
The point which he is making is that while cabon dioxide has little effect at the surface due to the level at which it exists being so much lower than that of water vapour and whatever effect it would have is swamped by the effects of water vapour which in essence absorbs and re-emits all of the infrared radiation there is, at the higher altitudes the atmosphere is very dry. As such carbon dioxide stands a far better chance of absorbing and re-emitting the infrared radiation.
Mike then focuses on what he regards as the more essential issue. When the radiation which is emitted by the carbon dioxide at the ground warms the surface, it will cause water to become warmer, leading to evaporation. Water vapor itself is a greenhouse gas and will absorb and re-emit further radiation, warming the surface a little more.
Continuing with this, as radiation rises in proportion to the temperature to the fourth power, the rise in temperature which results from each additional amount of infrared radiation emitted by additional water vapor will always be less than that of the previous amount of water vapour. This is why there is no runaway effect. While there is a feedback loop leading to a little more water vapour each time, the rise in temperature resulting from even an equal amount of water vapour will be smaller, and in fact less and less water vapour will be added as the process continues. Beyond a certain point, the added water vapour becomes negligible and the process effectively comes to an end.
But continuing with Mike’s point, it is the additional amount of carbon dioxide in the upper atmosphere which gets the whole process going in the first place. This is what it means for the additional carbon dioxide to be a “forcing,” whereas the additional water vapour is only a “feedback.”
*
In any case, all of this has undoubtedly been said many times in the past, and rob has probably heard it plenty of times before. But for the benefit of anyone new, it might help to repeat it again. But there really isn’t any reason why the same people have to go into depth every time.
Re #372: Thanks, Ray, my background is first year university science a while ago, and your explanatin does help. Let me try to restate it in simpler language.
A greenhouse gas molecule has two states of relevance to climate – the vibrational state affected by longwave radiation, and the kinetic state which is the temperature of the gas. The equipartition theorem implies the vibrational state can be induced by the kinetic state (ie. by molecular collision), so it is not something that only radiation could induce, as I had thought. A given input of longwave radiation to a part of the atmosphere will be partly re-radiated, and partly increase the kinetic energy and therefore the local temperature.
But if the atmosphere is in equilibrium there should be no net loss of radiative energy to warming the neighboring air molecues. And if the greenhosue gas molecule re-radiates quickly there will not be much time for kinetic energy transfer. So, it appears there will be no significant warming of the atmosphere by this method. The radiated photons do the warming. Half of the emitted photons will go up, and half down. Most of them will be absorbed by other greenhsoue gases, but eventually they either warm the Earth’s surface, or escape into space.
Let me test my understanding with this thought experiment. You have a mass of air in a virtual “box” at a certain temperature. Some of the kinetic energy will be converted into vibrational energy, and a photon will be emitted out of the box. Energy will be lost from the system, and therefore this mass of air is self-cooling. Of course, in the real atmosphere radiative and convective energy are also coming into the “box” so there is no cooling.
Now my last question, for now: How is the absorption and radiation of photons affected by their temperature? Does the Stefan-Boltzmann law have any effect in this case, even though a greenhouse gas is not a blackbody? I am trying to understand the validity of the idea that greenhouse effect is determined by the temperature of the gases that radiate their energy into space, which occurs in the upper atmosphere where carbon dioxide is relatively more abundant than water vapor.
Blair, one thing to keep in mind is that Stefan-Boltzmann is simply an empirical relationship written down around 1880 and states that the energy radiated by a blackbody is proportional to the 4th power of the absolute temperature.
For a non-blackbody, a term called the emissivity is included on the right side of the equation. A blackbody does not reflect any incident radiation, and the standard model is a box with a tiny hole in it. Black velvet is another.
For example, consider the difference in emissivity between sea ice and open water – sea ice will reflect 90% of incident sunlight, but open water reflects something like 10%. Thus, if sea ice melts due to warming currents, the change in emissivity acts as a positive feedback on the overall system, as it results in the ocean absorbing more incident solar radiation than before.
The blackbody radiation curve could not be explained by classical 19th-century physics, and this led to the origins of quantum mechanics: http://hyperphysics.phy-astr.gsu.edu/hbase/hframe.html
The quantum effect leads to discrete ‘quantized’ absorption bands in processes including rotational transitions (lowest energy), vibrational transitions (this is the infrared absorption region), electronic transitions (this is the visible-ultraviolet region), and beyond that are high-energy ionizations caused by x- and gamma rays.
A nice image of this is at http://brneurosci.org/spectra.png and also at http://www.iitap.iastate.edu/gccourse/forcing/images/image7.gif
This shows how O2+ozone absorb in the ultraviolet electronic region (which keeps skin cancer rates down) and CO2 and H2O absorb in the infrared vibrational region (leading to the greenhouse or blanket effect). See the ‘windows’? These are where gases like CFC’s play an important role in blocking outbound IR. This image seems to be for surface conditions – adding more CO2 at the surface will have little effect on IR absorption (see how the peaks are already at the 100% absorption level?)
ClimateAudit links to a chart that shows the effect of increasing CO2 at ground level, and they deceptively claim that this proves adding CO2 has no effect: here is the image – http://home.casema.nl/errenwijlens/co2/co2_absorption.gif
If you look at gases at the planetary surface, conditions are warm and dense enough that the rotational bands all overlap due to rapid molecular collisions, and you get a smooth infrared absorption curve. However, in the colder and lower pressure high atmosphere, there are fewer collisions and the smooth absorption curves take on a ‘picket fence’ appearance.
This can be seen for oxygen at http://brucegary.net/MTP_tutorial/OxyAbsSpec.png
– but here you are looking at discrete vibrational bands showing up in the electronic transition.
In the case of CO2, you will see discrete rotational bands showing up in the vibrational (IR) transition. To top this discussion off, here is a graph showing the atmospheric infrared absorption for the atmosphere at sea level and at the 11 km level in the atmosphere:
http://www.atmos.washington.edu/1998Q4/211/absorption.gif
Notice how the IR bands are NOT saturated at the 11 km level? Adding more CO2 at this level means more infrared absorption at this level. This (I believe) is also what leads to the logarithmic response of CO2 forcing to increased CO2, rather than a linear response – which is why doubling the CO2 from 200-400 ppm has the same forcing effect as going from 500-1000 ppm. (I sure hope I’ve got that right this time!)
To calculate what the effect of that is on the planetary surface temperature, you need to go into a radiative transfer model, and from there to a radiative-convective model for the atmosphere, and then couple that whole business to a ocean-land-cryosphere model. That’s what climate modelers have been up to for the past 50 years or so. They use observational and paleoclimate data to compare the models to – (and sometimes they don’t appreciate the difficulty of getting that data, especially from the oceans!)
But never mind all that, global warming is a big hoax…
Timothy (376), a quick clarification while I mull the above posts: are you saying that water vapor is not a forcing mechanism, period, or that its forcing properties are weaker than CO2 and its own feedback effects?
“For example, consider the difference in emissivity between sea ice and open water – sea ice will reflect 90% of incident sunlight, but open water reflects something like 10%. Thus, if sea ice melts due to warming currents, the change in emissivity acts as a positive feedback on the overall system, as it results in the ocean absorbing more incident solar radiation than before.”
The water vapor temperature of ice is much lower than water. During the day the sea, but not the ice is heated. This meansthat both the air temperature and the air pressure are greater over the sea, than they are over the ice, causing the wind to blow from the sea to the ice.
During the night both the sea and ice radiate IR, the sea has a halo of water vapor above it and so remains warm, at the dew point the water vapor falls as liquid water. Over the ice, there much less water vapor. On cooling the drop in air pressure and temperature causes the water vapor in the air to undergo a phase transition and to fall as water-ice crystals. Thus the change in water vapor pressure acts as a positive feedback system, water vapor is converted to snow and so the vapor pressure above the ice is always less than over the sea. There is a net transfer of water from the liquid sea to the soild ice.
——————————————————————–
Both paragraphs are true, you have positive feedbacks on both systems.
Blair, Ike did a really great job of outlining the atomic physics behind the effect. In terms of the thermodynamics, think of it this way. Start with the atmosphere at equilibrium. Now, consider IR radiation from Earth rising through the atmosphere. Initially in the dense, wet atmosphere near the surface, the mean free path is very short, and the IR photons are absorbed quickly, exciting vibrational and rotational modes in H2O and to a lesser extent CO2. The excited molecules can relax in different ways. They can collide with other molecules, and impart thermal energy to them–that keeps the energy in the lower atmosphere. They can re-radiate back toward Earth. That also keeps the energy in the lower atmosphere. Or they can re-radiate back toward space. Now the new IR photon is still in the lower atmosphere, so the process repeats many times. Eventually, however, some IR photons make it to the colder, drier, thinner upper atmosphere. Here the mean free path is longer, and molecules are more likely to escape. Also, because the atmosphere is cold here, the density of IR photons is lower than it is in the lower atmosphere. The lower atmosphere is like being under the covers in bed, while the upper atmosphere is the cold room around the bed. The net flux of photons is outward. OK, now we add more CO2. It doesn’t have much effect in the lower atmosphere. However, in the upper atmosphere, the effect is like throwing on a second blanket on a cold night. More photons get absorbed, and the density of photons increases, as more are being re-emitted or there are more collisions with excited CO2 molecules, and the average kinetic energy (and so temperature) goes up. The net flux of photons outward decreases because more are captured and then re-emitted back toward Earth.
Rod B., Please look at the plots Ike refers to. They are very illustrative of what is going on. I think that what Gavin and Tim mean is that the CHANGE in H20 is a feedback. In other words, the amount of water and therefore IR absorption increase because the temperature rises in the lower atmosphere–further adding to the warming. Gavin or Tim, correct me if I am wrong here.
Re #377
Blair, your post has raised some very interesting points. First you wrote:
A greenhouse gas molecule has two states of relevance to climate – the vibrational state affected by longwave radiation, and the kinetic state which is the temperature of the gas. The equipartition theorem implies the vibrational state can be induced by the kinetic state (ie. by molecular collision), so it is not something that only radiation could induce, as I had thought.
However, on a search with Google for “Equipartition Theorem” I found this web page from Manchester University: http://theory.ph.man.ac.uk/~judith/stat_therm/node81.html where it says “If this is not satisfied, the heat capacity will be reduced, dropping to zero at low temperatures. The corresponding degree of freedom is said to be frozen out; this is the situation for the vibrational degrees of freedom at room temperature.” In other words, the equipartion theorem does not apply to vibrational excitation in the troposphere which is at a temperature of or below that of room temperature. So the equipartion theorem cannot be used to justify the current models.
Then you wrote:
But if the atmosphere is in equilibrium there should be no net loss of radiative energy to warming the neighboring air molecues. And if the greenhosue gas molecule re-radiates quickly there will not be much time for kinetic energy transfer.
The base of the atmosphere is not in equilibrium. It continually changes during the day and night. Moreover, the time for vibrational relaxation is around 10^-6 seconds whereas the time betwen collisons at room temperature is 10^-10secs. That means that the excited molecules receive around 1000 collisons before they have time to lose their photons. Nearly all the collisons are with air molecules which cannot re-emit the photons.
Next you concluded “… in the real atmosphere radiative and convective energy are also coming into the “box” so there is no cooling.” This id true in most of the atmosphere, but at the top of the atmosphere, radiation escapes to space helping to balance the incoming solar radiation. There the air is cooled and descends to be replaced with warmer air from below. At the base of the atmosphere the reverse is true, with more radiation coming in and being absorbed, so that the air there warms.
You then, for a second time, questioned whether the Stefan-Boltzmann Law applied to greenhouse gas emissions.
Ike #378 has finally replied that it is only an empirical law, however that is not entirely true. “The law was discovered experimentally by Jožef Stefan (1835-1893) in 1879 and derived theoretically, using thermodynamics, by Ludwig Boltzmann (1844-1906) in 1884.” http://en.wikipedia.org/wiki/Stefan-Boltzmann_law NB. the law applies to blackbody radiation emanating from the conductive shells of the atoms of a solid or liquid. It does not apply to gases.
HTH,
Cheers, Alastair.
One denialist argument is that it is the sun, not CO2. Mechanisms are: Cosmic Rays might induce cloud nucleation which could increase reflectivity which would lower temperature. Cosmic rays reaching earth are deflected (reduced) by increased solar geomagnetic activity. So increased solar geomagnetic activity would increase temperature via this hypothetical mechanism. Increased solar irradiance would increase earths temperature directly. The following abstract seems to be consistent with this denialist view. The direction and magnitude of the temperature changes assigned to the mechanisms, 0.1 to 0.7 C, could be of the same order as those observed say as reported by AR4, depending on the length of time (not given in the abstract) associated with the change. I only have the abstract currently. What am I missing?
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 112, D11109, doi:10.1029/2006JD007864, 2007
Atmospheric temperature responses to solar irradiance and geomagnetic activity
H Lu, Jarvis, Graf, Young, Horne
Abstract
The relative effects of solar irradiance and geomagnetic activity on the atmospheric temperature anomalies (T a) are examined from the monthly to interdecadal timescales. Geomagnetic Ap (A p) signals are found primarily in the stratosphere, while the solar F10.7-cm radio flux (F s) signals are found in both the stratosphere and troposphere. In the troposphere, 0.1â??0.4 K increases in T a are associated with F s. Enhanced F s signals are found when the stratospheric quasi-biennial oscillation (QBO) is westerly. In the extrapolar region of the stratosphere, 0.1â??0.6 and 0.1â??0.7 K increases in T a are associated with solar irradiance and with geomagnetic activity, respectively. In this region, F s signals are strengthened when either the QBO is easterly, or geomagnetic activity is high, while A p signals are strengthened when either the QBO is westerly, or solar irradiance is high. High solar irradiance and geomagnetic activity tend to enhance each other’s signatures either making the signals stronger and symmetric about the equator or extending the signals to broader areas, or both. Positive A p signals dominate the middle Arctic stratosphere and are two to five times larger than those of F s. When solar irradiance is low, the signature of A p in T a is asymmetric about the equator, with positive signals in the Arctic stratosphere and negative signals at midlatitudes of the NH stratosphere. Weaker stratospheric QBO signals are associated with high A p and F s, suggesting possible disturbances on the QBO. The signals of A p and F s are distinct from the positive temperature anomalies resulting from volcanic eruptions.
Received 21 August 2006; accepted 26 February 2007; published 7 June 2007.
[Response: First off, the signals seen here are related to the stratosphere (not the surface) and are only related to the 11yr solar cycle. This follows on from previous work where 11 yr signals have been seen (and modelled) in stratospheric ozone and related properties. This has nothing to do with long term trends (which are non-existent in the solar proxies in recent decades) or the surface temperatures for which there is little to no evidence of solar cycle influence. – gavin]
Rod B (#379) wrote:
As I understand it, when calling water vapor a “feedback” rather than a “forcing,” the difference is a matter of degree. They are two ends in a spectrum. If we were to start pumping a great deal of water vapor into the atmosphere, there would be justification for considering that a “forcing,” but it would be quite temporary as the excess water vapor would tend to precipitate out of the atmosphere.
And it is just this residence time which Gavin focuses on in the following:
6 Apr 2005
Water vapour: feedback or forcing?
https://www.realclimate.org/index.php/archives/2005/04/water-vapour-feedback-or-forcing/
Water vapor has a residence time of a few days (roughly ten) whereas the residence time for methane is roughly a decade and that for carbon dioxide is decades to centuries. Anyway, the article goes into a bit more than just residence times and equilibriation. For example, the “instantaneous effects” of different greenhouse gases are not strictly additive. The combined effect of two or more greenhouse gases is always less than the sum of the effects of each greenhouse gas considered in isolation. Pretty obvious – but it is nice to see some numbers.
Quick aside: I keep being surprised by how much information this website has in it. Just mining some of the comments from the time that I have been seeing here would prove quite valuable – for me personally.
Ike (378), a minor clarification: I thought open water reflection varied from near 100% to 10% depending on the incident angle. Not true???
Folks, there are two different explanations being offered here, piecemeal, in response to people’s questions and assertions of belief –Alastair’s and the other one. And there are arguments about facts and theory between Alastair and the other people who are offering explanations.
This is really, really confusing for new readers. No one’s yet even answered DocMartyn’s question about how we can prove that the level of CO2 is increasing since the annual increase is less than the annual variability (I tried by asking, if I gain 12 pounds every winter and lose 11 pounds every summer, how can I not gain weight, but that’s argument by analogy).
Any chance of putting Alastair’s theory in a topic of its own as a parallel world kind of explanation So we could get say Ray’s and Gavin’s answers in a thread that assumes at basis the climate models discussed, and in the parallel world we could get Alastair’s answers where he could explain the basis for his ideas?
Getting them all mixed up must be endlessly amusing for those who believe this is impossible to understand.
But it needs to be understandable.
Editor, editor?
>378, 385
Rod B, when you’re talking about melting polar ice, remember the sun angle is constrained by the location.
Rob,
I had gotten this wrong:
(from #384)
It turns out, for example, that the “instantaneous” effect (that is, without a process of equilibriation where by the quantities of these gases vary over time) of carbon dioxide and water vapor (in terms of radiative forcing) is actually greater than the sum of their individual effects.
For the benefit of the non-specialists, I would like to explain why I thought it was the reverse, then move on to why it turns out to be otherwise. If one is considering both gases together distributed evenly within a single shell, since whatever radiation is absorbed by one is radiation which cannot be absorbed by the other, it would make sense that the presence of either will add to their combined effect, but will do so partly at the expense of the individual effect of the other.
But there are a number of problems here. The most obvious is that it is a drastic oversimplification: while water vapor acts primarily in the troposphere near the surface, carbon dioxide acts principally in the stratosphere, so even at this point the claim that some of the effect of one will be lost at the expense of the other begins to look questionable.
But far more importantly, the uniform shell analysis which I suggested leaves out one very basic fact: the greenhouse effect is itself a form of positive feedback. The radiation absorbed and re-emitted by the ground (where thermal energy enters the system) is absorbed and re-emitted by the atmosphere, which can then be absorbed and re-emitted by the ground – or absorbed and re-emitted by different parts of the atmosphere or even different gas constituents.
It is true that the absorbtion and re-emission of infrared by water vapor in the troposphere takes place at large expense to carbon dioxide – at that level. But in large part this simply has to do with there being so much more of the water vapour. But the feedback between the troposphere (which is where water vapor principally acts and the stratosphere (which is where carbon dioxide principally acts) is quite considerable – even before one takes into account any positive feedback loop which changes the level of either gas.
*
Now what can we conclude from this – besides the fact that I am embarassed?
Things can get a little complicated – enough that what seems like it is just commonsense isn’t necessarily right. I was looking at one aspect of the problem, but not at others – even though I knew about them. Commonsense will get you only so far, especially when dealing with some of the more advanced science topics. And as a matter of fact, oftentimes the specialists will simply solve the equations rather than look for the “story” that helps people like myself understand, where by “story,” I mean an explanation which tries to separate things and looks at how they interact at a more or less qualitative level – and which can be more easily compared to or is in some way more or less analogous with things which we know from everyday life.
Now this does not mean that non-specialists should give up trying to understand the more esoteric theories.
That would be the last lesson anyone should ever take away from this. But what it does mean is that when someone like myself puts some time into trying to understand such things, this should be done with a fair amount of humility – and with the recognition that some people have put a great deal more time into studying such things than I have. In any disagreement, if you had to lay down money, placing it on the specialist will probably be a safe bet. In the meantime, if a layman such as myself takes an interest in a given field and does so with a certain amount of humility, the specialist will probably take it as a compliment. When people aren’t too busy, they will often enjoy sharing their mutual interests.
One other point: if there is any complexity to a given issue, it is probably better to say that something “seems obvious” rather than that it “is obvious”!
Re #385
Ike was right about perfect blackbodies emitting at the same rate as they absorb, but that is only true when they are in a state of thermal equilibrium, for instance in an isothermal cavity. That is not the case for the Earth, since the Sun radiates at a much higher temperature than the Earth’s surface.
Moreover snow and water are not perfect blackbodies. While snow is highly reflective at the wavelength of visible light, it behaves like a blackbody at 0C in the infrared wave bands. And, as you pointed out the albedo of water varies with the angle of inclination of the sun, and its surface roughness, but it also emits in the infrared with a spectrum close to that of a blackbody, unlike water vapour which emits bands of lines.
Both snow and water absorb infrared radiation with a strength close to that of a blackbody, but that is only important when there is cloud cover, since clouds emits as blackbodies in the infrared too.
HTH,
Cheers, Alastair.
Re#381, Ray that’s a very helpful description of how the process works in the atmosphere. In particular, the phrase “The net flux of photons outward decreases because more are captured and then re-emitted back toward Earth.” helps with understanding a surprising phenomenon.
What I always found quite surprising was that if you measure the temperature of Venus from space, it appears colder than Earth! This is true even though surface temperatures on Venus will melt lead.
With respect to incident sunlight – for Venus, the flux at the top of the atmosphere is 2643 watts/square meter, and for Earth it is 1370. Venus has an albedo of 0.8, and Earth has an albedo of 0.3, meaning that the absorbed solar radiation at the surface is actually less for Venus than it is for Earth.
The surface temperature of Venus is 457C, and for Earth it is 15C. However, if you take a temperature reading from space, you will get -53C for Venus and -18C for Earth.
The difference between these values is the ‘greenhouse effect’ – which is 510C for Venus and 33C for Earth. (Venus has an atmosphere that is 97% CO2).
This remarkable result is the reason why stratospheric cooling is predicted to result from an enhanced greenhouse effect on Earth. It seems clear that if increased solar radiation was responsible, you’d see stratospheric warming. See the RC article, The sky IS falling.
RE Alastair’s comments: Nowhere did I state that the Earth is a perfect blackbody. The issue of concern is the wavelength dependence, i.e. the emissivity. Again, see the figure http://www.atmos.washington.edu/1998Q4/211/absorption.gif The top line shows what blackbody curves look like; the bottom is the atmospheric absorption.
Are you now willing to admit that your novel theories about longwave emission are incorrect?
Alastair, the atmosphere is essentially transparent to the sun’s radiation which is why one talks about a local thermodynamic equilibrium, which is why you can use Kirchhoff’s law.
“No one’s yet even answered DocMartyn’s question about how we can prove that the level of CO2 is increasing since the annual increase is less than the annual variability (I tried by asking, if I gain 12 pounds every winter and lose 11 pounds every summer, how can I not gain weight, but that’s argument by analogy).”
I have not asked that question. I know the level of CO2 is rising. I trust the CO2 levels recorded world wide. The question I asked was about RATES of change. Kinetics is about rates. So cut out the strawman arguments.
>392
See 255; what do you mean by “steady state” — linear increase?
“Is there a reason for the variablity of data sets in the graph (1) above in the time periods between 800-1800?? It seems as though all of the data in more recent years is much more uniform. Why is this? For example DWJ2006 and ECS2002 appear to vary in measurements by as much as .5C or more in some cases versus JBB1998 and MJ2003. Even if they are using different models or proxy data why would they all begin to move in lock step as time moves closer to present? Is the data set just more accurate now?”
Can someone please answer this?? Or send me a link at least where I can get an explanation? I thought this was a site for dicussion of climate science…
Hank, here is an explanation of steady state, Wikpedia has a go with chemical steady states, but there are no good biological ones in there.
The term steady state describes a situation where some components, but not necessarily all, of the dynamic system are constant. The system is dynamic; so there is overall flux through the system, but some parts of the system stays the same. Many systems behave in this same manner, the level of a non-tidal river will remain more or less constant during the course of a day, even though water is gushing into the sea down stream. Hydroelectric dams are kept at steady state by allowing feed waters into the reservoir to be varied so as to maintain a static head, even though changes are made to electricity, hence water, demand.
A person weight is also a steady state, although you put on weight after a meal and loses weight after a trip to the bathroom; there weight fluctuations describe the maximum changes in the steady state that is possible. It is true that one can gain a lot of weight by eating a large meal and drinking a few liters of water, but this change of up to 4 kilo�s per hour does not describe the maximum rate at which you can change your steady state rate. You cannot lose body weight by spending more time in the bathroom. The analogy of a person�s weight and the level of atmospheric CO2 is a good analogy. A person has various weight inputs, meals, snack and drinks and loses weight by excretion of waste produces. You can slowly increase your weight by having inputs, calories, higher than your outputs. You gain weight by not using your muscles and by having a high calorific input.
Same with atmospheric CO2, there are a large number of inputs into the atmosphere and a large number of outputs, add them together and you get a steady state. The CO2 readings from Hawaii show that the RATE of change in CO2 between April and October and then October and April are greater than the slow, slight increases in CO2 seen over decades. As the steady state changes are very slow compared with the six-monthly changes, we know the system is at steady state.
You can also have steady state systems where none of the component are static, but the overall systems is a steady state. Menstruation is a good example of this, during a menstrual cycle no components are at steady state, but the changes in follicle-stimulating hormone, luteinizing hormone, Estrogens and gonadotropin releasing hormone are at steady state temporally, i.e. if you only measured them every 28 days they would be the same. The whole of the �system� is at steady state, but not the components.
Once you realize that the system is in steady state, and not an equilibrium then you can do some maths and look at some of the system dynamics. You can ask the question, where does all the CO2 go? Every year geological action releases about 0.5 GT of CO2 into the atmosphere. It did this before humans evolved and will do it after we are extinct. As pre-industrial CO2 stayed at about 280 ppm for a long time, we must look for a mineralization pathway that gets rid of 0.5 GT per year. If it is a cation/carbonate process, it might be nice to know something about the order of the reaction.
>steady state
Do these illustrate what you’re describing?
http://www.globalwarmingart.com/wiki/Carbon_Dioxide_Gallery
Simply stated, a steady state is a situation where the rate of loss equals the rate of gain for something. In chemistry this is usually an intermediate that is produced in one reaction and consumed in another. In such a situation there is no change. This describes the situation for CO2 before 1850 or so. However, people have added another source term, burning of large amounts of fossil fuel. Getting rid of the accumulated excess by natural mineralization will require geological time scales. Tens of thousands to millions of years. Using chemical or physical methods for mineralization requires significant energy, which we would get by?
deja – all the reconstructions are constrained by the same global instrumental record which starts about 1850. The reconstructions are calibrated using the same instrumental record thus they converge to it and each other at about 1850.
Simply stated, a steady state is a situation where the rate of loss equals the rate of gain for something. In chemistry this is usually an intermediate that is produced in one reaction and consumed in another. In such a situation there is no change. This describes the situation for CO2 before 1850 or so. However, people have added another source term, burning of large amounts of fossil fuel. Getting rid of the accumulated excess by natural mineralization will require geological time scales. Tens of thousands to millions of years. Using chemical or physical methods for mineralization requires significant energy, which we would get by?
deja – all the reconstructions are constrained by the same global instrumental record which starts about 1850. The reconstructions are calibrated using the same instrumental record thus they converge to it and each other at about 1850.
Hank Roberts (#396) wrote:
The first one looks an aweful lot like what he is calling “steady state.” Its even got the wiggles. Oddly enough, it doesn’t seem at all like what the material he was quoting from Wikipedia was refering to, though.
Re #381: Ray, thanks for your response. You said
You are describing two separate warming processes. Again, I would like to know the relative significance of the transfer of thermal energy directly to the atmosphere. The greenhouse gas models I have read about (including Gavin’s Learning from a Simple Model) do not mention it, they only talk about IR radiation warming the ground. You then say
I thought the density of photons was lower because they had been absorbed lower in the atmosphere and converted to thermal energy, whether on the ground or in the air. If the temperature has anything to do with how a greenhouse gas emits IR radiation, I would like to know what it is.
Re #378: Ike, thanks for the information of the broadening of the rotational water vapor bands under atmospheric pressure. I would thus assume that the graph of overlapping water vapor / carboin dioxide aborption bands on this NASA page is misleading. They do not say where this was measured – at the surface or is it an average of the entire atmospere.
Re #388: Timothy, you state that the effect “of carbon dioxide and water vapor (in terms of radiative forcing) is actually greater than the sum of their individual effects, but I do not agree with your explanation of this statement. I do not thing what happens after the photon is aborbed is relevant to the question. The effect is logarithmic, not additive – it does not matter if you add another unit of water vapro or carbon dioxide.
I do share with you the desire to come up with a qualitative “story” of how the greenhouse effect works based on the physical processes involved.. This is different than Gavin’s simple model which emphasises the mathematics. I am learning a lot of pieces to the puzzle, but a coherent picture remains elusive.