According to some recent reports (e.g. PlanetArk; The Guardian), the public concern about global warming may be declining. It’s not clear whether this is actually true: a poll conducted by researchers at Stanford suggests otherwise. In any case, the science behind climate change has not changed (also see America’s Climate Choices), but there certainly remains a problem in communicating the science to the public.
This makes me think that perhaps a new simple mental picture of the situation is needed. We can look at climate models, and they tell us what we can expect, but it is also useful to have an idea of why increased greenhouse gas concentrations result in higher surface temperatures. The saying “Everything should be made as simple as possible, but not simpler” has been attributed to Albert Einstein, which also makes me wonder if we – the scientists – need to reiterate the story of climate change in a different way.
Gavin has already discussed this (also see here and here), but it may be necessary to tell story over again, with a slightly different slant. So how can we explain how the greenhouse effect (GHE) work in both simple terms and with a new angle? I also want to explain why the middle atmosphere cools with increasing greenhouse gas concentrations associated with an increased GHE. Here I will try to present a conceptual and comprehensive picture of GHE, explaining both the warming in the lower part of the atmosphere as well as the cooling aloft, and where only the most central features are included. Also, it is important to provide a good background, and we need to start with some very fundamental facts.
Four main physical aspects
Several factors are involved, and hence it may be useful to write a simple recipe for the GHE. This recipe then involves four main ingredients: (i) the relationship between temperature and light, (ii) the planetary energy balance, (iii) the distance light travels before being absorbed, and (iv) the relationship between temperature and altitude.
(i) Temperature and light
Energy can be transmitted in many different ways, involving photons (light or electromangetic radiation), conduction, and motion. Most of these require a medium, such as a gas, fluid, or a solid, but space is basically a void through which photons represent virtually the only form for energy transfer. Hence, planets tend to gain or lose energy to space in the form of photons, and we often refer to the energy loss as ‘radiative heat loss’.
A fundamental law of physics, known as the Planck’s law, says that radiative heat loss from any object depends on its temperature. Planck’s law also explains the colour of the light, or its wavelength, and hence explains why iron gets red hot when heated sufficiently.
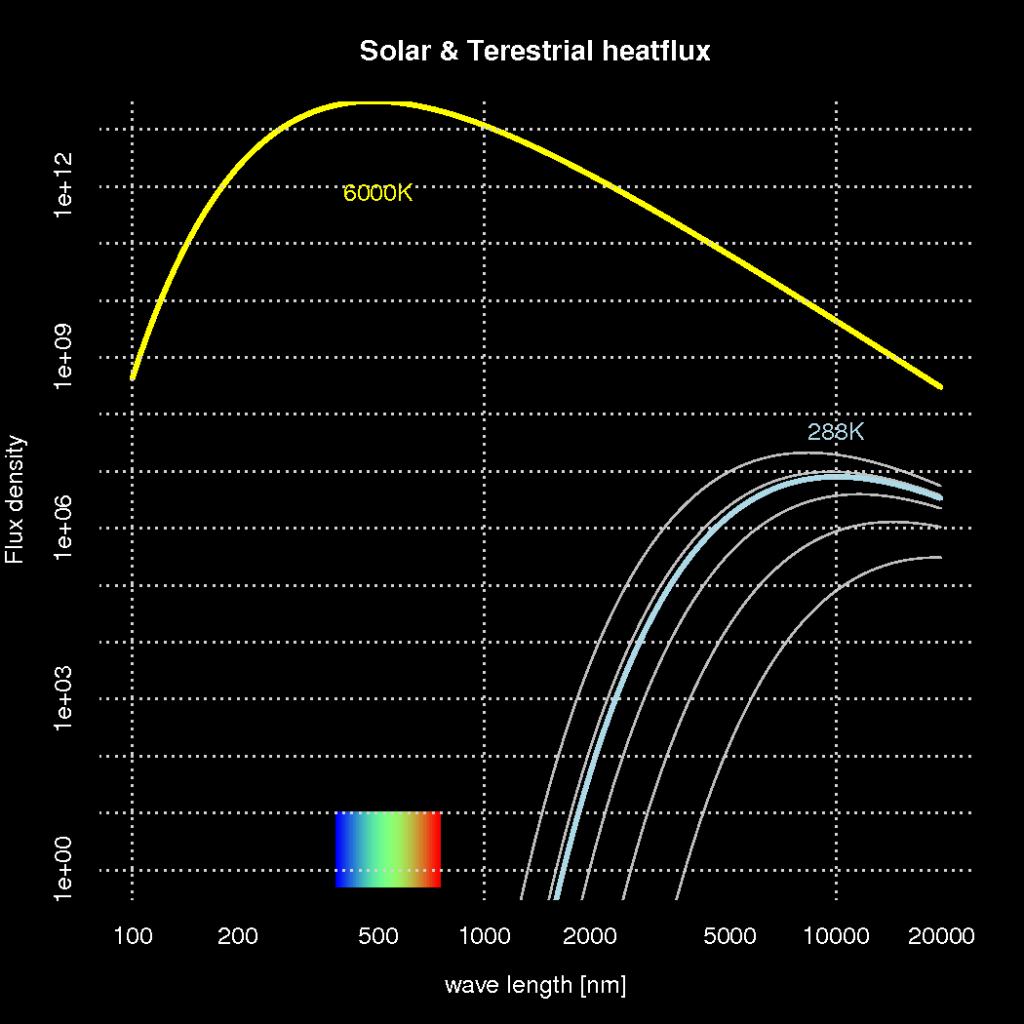
Planck’s law predicts that the light from an object with a temperature of 6000K – such as the solar surface – produces light that is visible, whereas objects with a temperature of 288K produce light with a wavelength that our eyes are not able to see (infra red). This is illustrated in Figure 1 showing how the light intensity (y-axis; also referred to as ‘flux density‘) and the colour of the light (wave length) vary for objects with different temperatures (here represented by different curves). The yellow curve in the figure represents the solar surface and the light blue curve the earth.
(ii) The planetary energy balance
The planetary energy balance says that our planet loses heat at the same rate as it receives energy from the sun (otherwise it would heat or cool over time). This is because energy cannot just be created or destroyed (unless it involves nuclear reactions or takes place on quantum physics scales).
The planets’ distance from the sun and the brightness of its surface dictates how much energy it receives from the sun, as the light gets dimmer when it spreads out in space, as described by Gauss’ theorem.
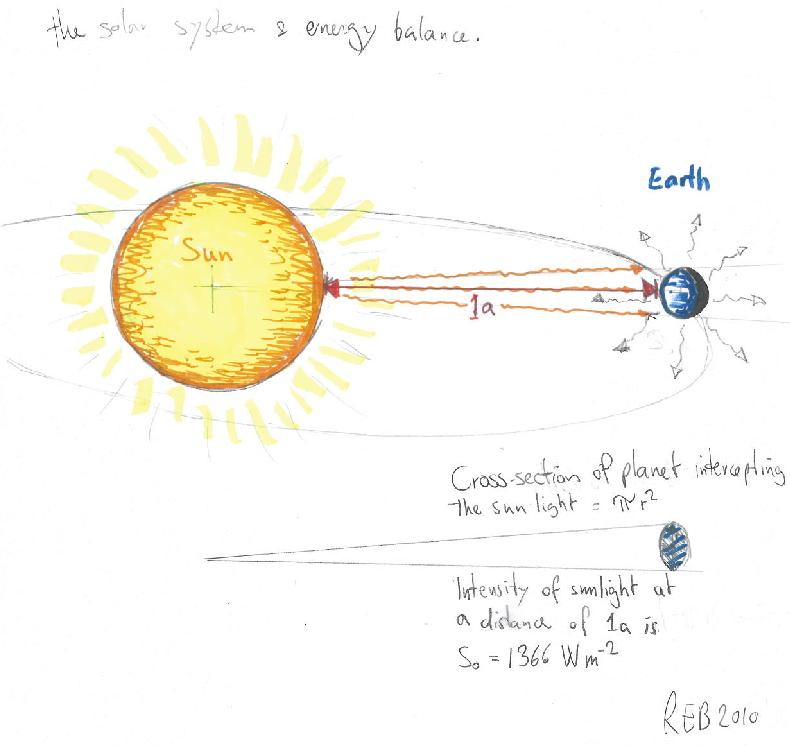
The energy flowing from the sun is intercepted by the earth with energy density described by the ‘solar constant‘ (S0=1366W/m2), and the amount of energy intercepted is the product between this flux density and the earth’s disc (minus the reflected light due to the planet’s albedo: A ~0.3). The average heat loss is given by the product of earth’s surface and its black body radiation:
S0/4 (1-A) = σT4,
where σ=5.67 x 10-8W/(m2 K4) is the Stefan-Boltzman constant. This gives a value of 255K, known as the emission temperature.
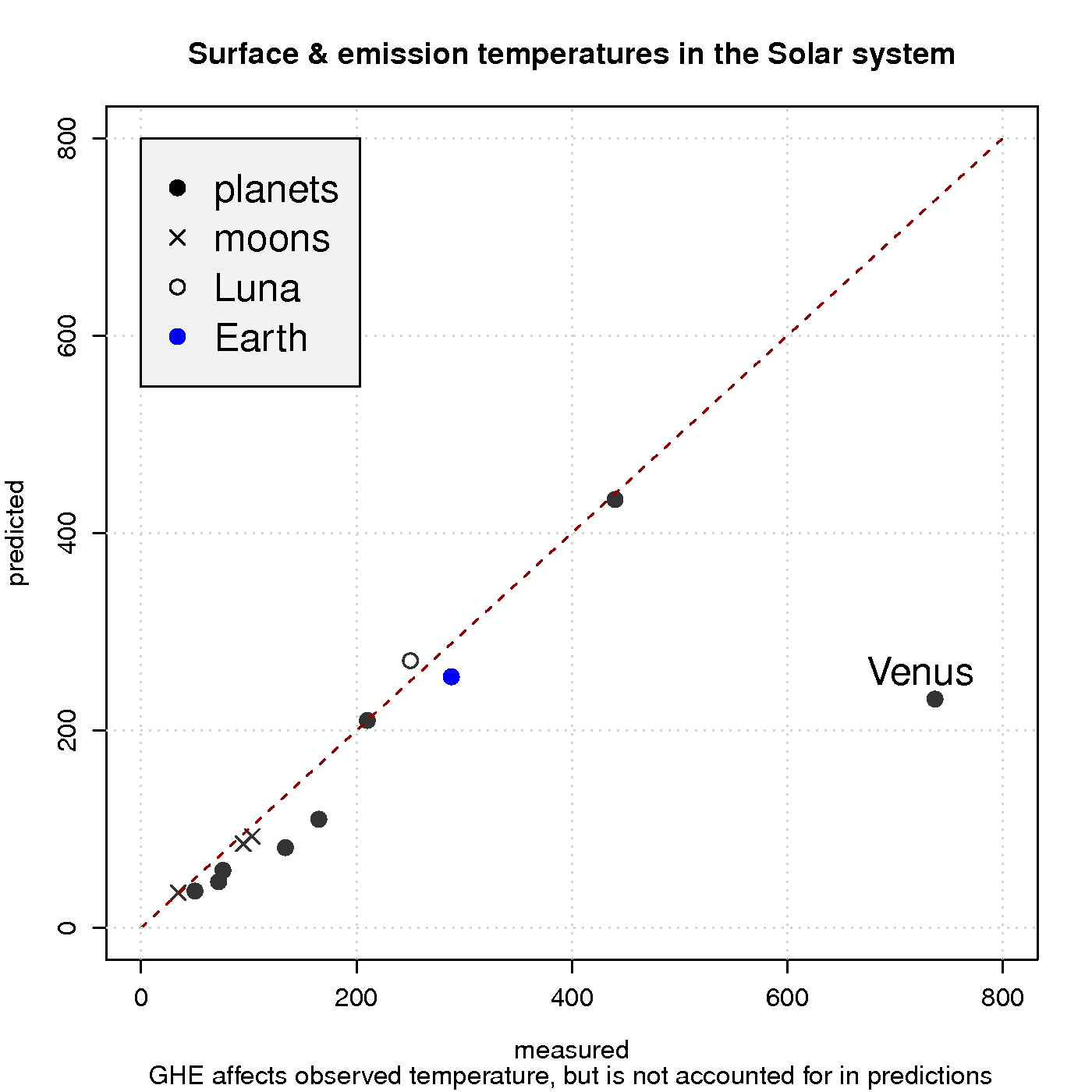
Figure 3 shows a comparison between observed surface temperature and calculated emission temperature for the planets in the solar system, based on the balance between energy from the sun and heat loss due to black body emission. In these simple calculations, the greenhouse effect is neglected, and the black body radiation can be derived from Planck’s law. The calculations agree quite well with the observations for most of the objects in our solar system, except for Venus which is known to harbour a strong GHE and has a hotter surface than Mercury despite being about twice as far away from the sun.
(iii) Light absorption
It is also clear that our planet is largely heated at the surface because the light from the sun – which is visible for our eyes – penetrates the atmosphere without much absorption (hence we can see the sun from the ground). However, the atmosphere is a medium of gas and particles that can absorb and scatter light, depending on their wavelength (hence explain why the sky is blue and sunsets orange).
The distance light travels before being absorbed – optical depth – can vary with the light’s wavelength and the medium through which is travels. The optical depth in our atmosphere is different for visible and infra-red light.
Infra-red light is absorbed by molecules, which in turn get more energetic, and the excited molecules will eventually re-emit more infra-red light in any random direction or transfer excess energy to other molecules through collisions. In a optically thick (opaque) atmosphere, there will be a cascade of absorption and re-emission.
Hence, whereas the planet is heated at the surface, it’s main heat loss takes place from a height about 5.5 km above the ground, where most of the radiation is free to escape out to space. The optical depth dictates how deep into the planet’s atmosphere the origin is for most of the planet’s infra-red light (the main planetary heat loss) that can be seen from space. Furthermore, it is the temperature at this level that dictates the magnitude of the heat loss (Planck’s law), and the vertical temperature change (lapse rate) is of course necessary for a GHE. The temperature at this level is the emission temperature, not to be confused by the surface temperature.
We know that the optical depth is affected by CO2 – in fact, this fact is the basis for measuring CO2 concentrations with infra-red gas analysers. Molecules composed of three or more atoms tend to act as greenhouse gases because they can possess energy in terms of rotation and vibrations which can be associated with the energy of photons at the infra-red range. This can be explained by theory and be demonstrated in lab experiments. Other effects are present too, such as pressure and Doppler broadening, however, these are secondary effects in this story.
(iv) The relationship between temperature and altitude
There is a well-known relationship between temperature and height in the troposphere, known as the ‘lapse rate‘ (the temperature decreases with height at a rate -6K/km). The relationship between temperature and altitude can also be seen in the standard atmosphere. The lapse rate can be derived from theory for any atmosphere that is the hydrostatically stable condition with maximum vertical temperature gradient, but it is also well-known within meteorology. Thus, given the height and value of the emission temperature, we can get a simple estimate for the surface temperature: 255K + 5.5km * 6K/km = 288K (=15oC; close to the global mean estimated from observations given by NCDC of ~14oC).
Enhanced greenhouse effect
The term known as the ‘enhanced greenhouse effect’ describes a situation where the atmosphere’s becomes less transparent to infra-red light (reducedincreased optical depth), and that the heat loss must take place at higher levels. Moreover, an observer in space cannot see the infra-red light from as deep levels as before because the atmosphere has become more opaque.
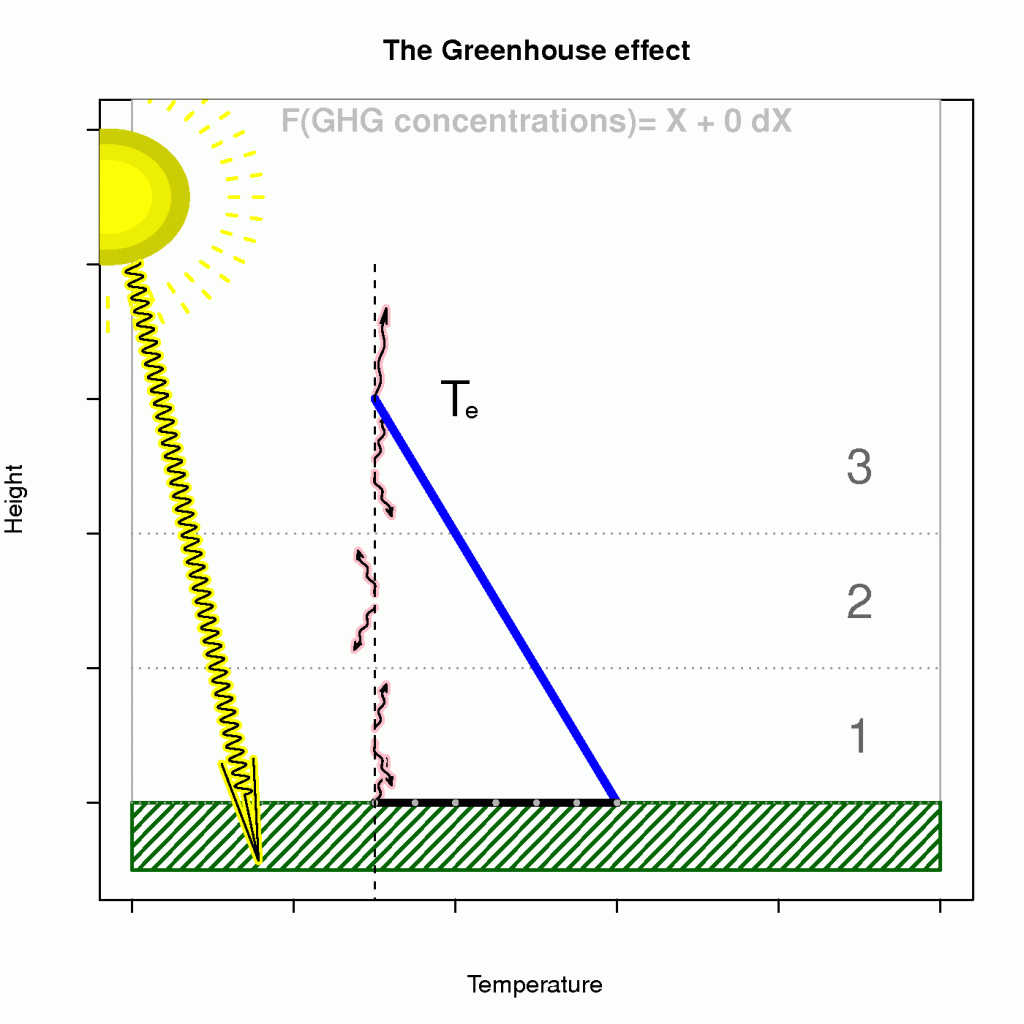
The effect of heightened level of heat loss on the surface temperature is illustrated in Figure 4, where the emission temperature and lapse rate are given if we assume an energy balance and a hydrostatically stable atmosphere on average (a generally hydrostatically unstable atmosphere would be bad news).
Hence, a reducedincreased optical depth explains why atmospheres are not easily ‘saturated‘ and why planets such as Venus have surface temperatures that are substantially higher than the emission temperature. Planets with a thin atmosphere and insignificant greenhouse effect, on the other hand, have a surface temperature that is close theto the estimates from the planetary energy balance model (Figure 3).
Feedback processes
The way the atmosphere reacts to changes in the optical depth is more complicated, due to a number of different feedback mechanisms taking place. But to get a simple overview, it is useful to keep in mind that the optical depth is sensitive to how much water vapour (humidity) there is in the air, and that the lapse rate is sensitive to the composition of the atmosphere (i.e. humidity). Furthermore, the albedo A is affected by clouds, snow, ice, and vegetation, all of which affect the way the earth receives energy from the sun.
In our simple picture, feedback processes affect changes in the height of the level where most heat loss takes place, the slope of the lapse rate, and heating at the surface (and hence the emission temperature).
So why is the upper atmosphere cooled then?
The upper atmosphere, comprising the stratosphere and mesosphere, is expected to cool during an AGW, as shown by the GCMs. So what is happening there? This is when the picture becomes more complicated.
Since CO2 mostly absorbs/re-emits infra-red light at around 14 microns, an increased concentration in the troposphere will reduce the upward infra-red radiation at this band. The total energy is roughly constant, but it is made up from increased emissions at other bands because it’s warmer. There is less absorption by CO2 of upwelling infra-red light above the troposphere, but increased emission as a function of increased concentrations. Thus there is a cooling.
Controversy?
Can this picture be falsified, e.g. if other factors were to play a role too? For instance, can this situation be altered by changes in the sun?
Changes in the sun can of course affect the amount of energy received by the earth through changes in its output, variations in the intensity of UV-light, or perhaps even clouds through galactic cosmic rays. But it’s hard to see any systematic long-term trend in the level of solar activity over the last 50 years, and it is difficult to see how solar activity may have an effect while other factors, such as GHGs, don’t. Gavin and I recently published a study on the response to both solar activity and GHGs, and found similar magnitude for both forcings in both observations and the GISS GCM.
There have been claims of negative feedbacks, such as the “iris effect“. One would expect negative feedbacks in general to dampen the response to most forcings, unless they involve a particular process that is active for a particular forcing. In other word, why would a negative feedback act for GHGs but not for solar forcing? Many feedbacks, such as changes in atmospheric moisture, cloudiness, and atmospheric circulation should be similar for most forcings.
Another question is why we do see a global warming trend if the negative feedbacks were most important (Figure 5). Negative feedbacks usually imply quiet conditions in a complex system, whereas positive feedbacks tend to lead to instabilities, often manifested as internal and spontaneous oscillations (see Figure 5). It is reasonable to expect the feedback processes to affect natural variations as well as forced changes such as an enhanced GHE, orbital changes, volcanoes, or changes in the sun.
The point about negative feedback also brings up another interesting issue: Negative feedbacks usually act to restore a system to a particular zero-level state. What would the zero-state be for our climate? No greenhouse effect or some preferred level of greenhouse warming? There is already a natural GHE that, together with other atmospheric effects, can account for about 32oC higher global mean surface temperature. What makes this state so special, and can we explain the present natural GHE in the presence of negative feedbacks (consider starting from a state with no GHE)?
Hence, claims of negative feedback is controversial because all these tough questions then need to be addressed. We can write down a simple recipe for the GHE, but it is indeed challenging to reconcile a presence of a negative feedback with our observations, or explain the current observed global warming in any other terms.
I recommend editing this article to take into account some of the comments that have been made here. I agree with ononymous that the sentence regarding conservation of energy and nuclear reactions is really really jarring. Also I agree with Chris Dudley that the discussion of optical depth is incoherent. For starters optical depth is not a distance. Second, optical depth increases with increasing CO2. If you are trying to convey some subtlety regarding optical depth and the enhanced greenhouse effect, it is opaque. This is not a journal article; it can be edited after publishing. I know that writing these things is a lot of work and I do appreciate the effort.
McGahill 177,
No, greenhouse gases don’t particularly retain heat. They absorb infrared photons, which heats them up, and they then emit heat.
McGahill (#177),
In addition to what’s been said already, the difference between O2 and CO2 (as an example) is that O2 is perfectly balanced while CO2 is not and that makes it excitable. Specifically, you might say that an electric field can bend the molecule by pulling at the carbon because it does not have the same charge as the oxygen.
If you want a full explanation, David Archer recorded accessible video lectures “for English majors”. Lecture 6 explains this stuff but I recommend you check lectures 2 and 3 first which explain the basics in detail (skip if you’re familiar with the material already of course).
Didactylos
“A good graph can explain a lot. This is the reason (probably the only genuine reason) why the hockey stick graph has been attacked so brutally.”
If and Only if people are able to interpret the graph. Most people do not know the difference between the y axis and the x axis. And the attacks on the hockey stick graph have been successful for this very reason. The average person does not have much mathematical training.
The general population, by that I mean soccer moms and football dads, needs different explanations than people with technical backgrounds.
[edit – OT topic, original comment removed]
To Scott Mandia (#196), who states: As mentioned here, as the troposphere becomes more optically thick, the height at which IR is released to space is higher and cooler which means there is less upwelling IR to the regions above the troposphere. So is this causing the stratospheric cooling? It appears that the answer is “yes, to some degree” but there are other factors that make it difficult to nail down this thing..
Scott – In a steady state (where the models predict stratospheric cooling just as they do under current forcing conditions), for the troposphere to radiate IR to space in an amount that balances the heat absorbed from solar radiation in the troposphere and at the surface, it must radiate at a temperature averaging about 255 K (that’s the temperature needed to reradiate the absorbed heat via the Stefan-Boltzman equation). That radiation doesn ‘t occur at any single altitude, but the “radiating layer” (i.e., the level with a 255 K temperature is a hypothetical altitude equivalent to the average of the multiple layers that actually radiate, some warmer, some cooler. The 255 K altitude rises with greenhouse effects, but the calculated temperature doesn’t change (unless the sun heats up or cools down or the albedo changes). In essence, greenhouse effects change the level of tropospheric radiation, but not the temperature or the amount of radiation.
To summarize the above, the amount of radiation leaving the troposphere to enter the stratosphere is fixed by the amount of heat the troposphere and Earth’s surface absorb, and is for practical purposes unaffected by CO2. Increased CO2 merely raises the average temperature below the radiating layers needed for that radiation to be emitted by those layers. In a circumstance of radiative imbalance, that scenario changes slightly, but the current imbalance is far too small to account for more than miniscule stratospheric temperature changes. Hence, one can’t explain stratospheric cooling mediate by CO2 on the basis of reduced IR radiation entering the stratosphere.
What appears to be a critical role of ozone in permitting added CO2 to cool the stratosphere by serving as an escape mechanism for ozone-mediated warming is discussed in 3, 14, 28, and 41. The last of these mentions other heat-absorbing moieties beyond ozone, but in the stratosphere itself, ozone is probably the most important one. Item #14 is the one I would most emphasize, simply because of the credentials of the geophysicist I quote on the subject, Raymond Pierrehumbert, whose forthcoming book will address this topic quantitatively in some detail.
Optical Depth is such a tricky factor, but the atmosphere becomes more or less opaque at various wavelengths, so when one adds CO2 it doesn’t mean it becomes more and more opaque at all IR wavelengths. Absorption varies with height and pressure as well.
http://aeronet.gsfc.nasa.gov/cgi-bin/type_one_station_opera_v2_inv2?site=Resolute_Bay&nachal=0&year=18&month=6&aero_water=0&level=2&if_day=0&if_err=0&year_or_month=0
Is complicated a bit, but not too confusing.
I am particularly interested in why the models have failed to predict Arctic sea ice volume going down so fast. I believe in extra IR downwelling responsible for more thawing and also CO2 becoming more prominent in dryer colder climates, but I have seen scant evidence in that area, and what I read mainly denies this, but it does make sense, the stratosphere cools due to lack of IR causing warming, ozone gets depleted further by a colder stratosphere. This is a great success as described by models. Yet sea ice extra depletion evades explanation.
I am also a novice at this one: sea ice CO2 permeability , wow, this may be something worth understanding more.
But all and all, the sun disks of the Arctic regularly expand in size almost every year like clockwork. 2010 has shown
very large expansions, surpassing all previous years, the only thing doing this is lesser air density, pressure usually varies quite a lot along a well defined median, so only thing left is temperature. Warmer and warmer we go, as the world already knows, but little do we do something about it.
The greenhouse effect can be explained by the urban heat island effect.
Imagine a house in a rural area, surrounded by cool vegetation, that will be comfortable on a sunny spring day.
Now imagine this same house, on the same day, but downtown in an urban area. In addition to the heat from the sun, it is also getting heated by Infrared radiation from the brick office building across the street, and the hot cars in the parking lot next door, and so on. The brick office building is made hotter by the Infrared radiation from the parking lot, and the parking lot is made hotter by the Infrared radiation from the office building; both are made a teeny bit hotter by the Infrared radiation from our little house. Some of the directions where our house used to lose heat by radiation now have warm objects in the way; our house is still radiating heat, but these objects are absorbing it, and radiating some of it back. As the neighborhood became built up, so did the total heat coming to our house. Convection, conduction, and latent heat transport move the heat around, but almost all the heat come in as visible radiation, sunlight, and it all (eventually) leaves the earth as Infrared radiation.
Adding more CO2 (which absorbs & reradiates Infrared radiation) to the atmosphere has a similar effect to building an urban environment – it blocks paths where heat used to escape by Infrared radiation, and because it’s warmed by this IR, it reradiates additional energy (infrared) which adds to the energy coming from the sun in the visible, making things warmer. Because the additional CO2 from everybody’s power plants, and trucks, cars, cargo ships, furnaces etc quickly mixes into the atmosphere, the warming effect isn’t confined to where the CO2 emission occurs. The heating caused by urbanization in New York or New Delhi is greatest locally; the heating caused by CO2 is global, affecting Arctic sea ice, Himalayan glaciers, Antarctic ice shelves, rainfall extremes, and a lot of other things.
http://motls.blogspot.com/2010/07/rasmus-benestad-negative-feedbacks-are.html
Motl has a rant about this article. He seems not to have heard of Einstein and E=mc²
“Mr Benestad apparently believes that both nuclear reactions and quantum mechanics violate the energy conservation law! Well, they don’t, Mr Benestad. As Emmy Noether has shown, the energy is conserved whenever the laws of physics are invariant under translations in time.”
177. Comment by McGahill ”
Excellent to have this review of the science of atmospheric warming for us who have not been fully blessed with a scientific education. One thing that I have never had explained and that I would very much like to understand is this: I believe carbon dioxide and methane are greenhouse gasses effectively because they retain heat more or longer than the Oxygen and Nitrogen that make up the bulk of the atmosphere. But why are these carbon gases more retentive of heat? I assume it is something to do with the electronic structure of the molecules and the quantum energy levels in the molecules? Can someone please givean explanation sufficiently simple for a non scientist to grasp.”
————–
To put it simply, O2 and N2 are symmetric diatomic molecule that, while they have stretching like a spring, and can spin around, these motions to not possess a dipole moment between quantum states, and, hence, they cannot interact with an electro-magnetic field. Radiative process require EM field interations with dipole moment transitions.
Tiatomics quantum states of CO2, N2O, O3, H2O, etc, do have dipole moments which can interact with EM waves, so they can absorb and emit photons at frequencies (determined by quantum mechanics) corresponding to energy level differences between the various states they can assume. An EM transition is permitted if there is a net dipole moment change between the states. It’s complicated, I know, but it’s How God Made It. CO2 is a particularly strong infrared active molecule which is also notoriously chemically stable.
Re 185 Gordon says Climate change, and its anthropogenic source, directly challenges the “heroism” of western culture. To it up will be to give up the dream of immortality for billions of people. All the careful explanation in the world will not help.
Or the hero could be a dynamic character, who drastically reduces his/her reliance on fossil fuels, etc, and triumphs over AGW.
Re 201 John E. Pearson If you are trying to convey some subtlety regarding optical depth and the enhanced greenhouse effect, it is opaque.
Nice pun!
Re 177 McGahill – GHGs or other GH agents (clouds) affect the flow of radiant heat in somewhat the same way that replacing a layer of aluminum with a layer of cork affects the conduction of heat through the layer.
Re myself Re 177 McGahill – GHGs or other GH agents (clouds) affect the flow of radiant heat in somewhat the same way that replacing a layer of aluminum with a layer of cork affects the conduction of heat through the layer.
(Actually, they can do the opposite in some cases – ie they can increase the heat transfer over shorter distances, until the optical thickness becomes sizable over such distances; then farther increases will act like insulation. Basically, the average distance the photons can travel from emission to absorption is on the order of unit optical thickness (depending on the relative roles of absorption verses scattering; LW scattering is relatively minor for Earthly conditions); they can travel as easily in one direction as they can in the opposite direction, so an imbalance in photon flow – a net flux of photons – will be directed from warmer to colder regions, depending most on the variation in temperature over the spatial scale of unit optical thickness. Changing the optical thickness relative to the temperature distribution will change the net photon flux. Energy accumulates or is depleted depending on convergences or divergences of fluxes; the climate tends to adjust to bring the fluxes into balance.)
Patrick #66>> But here is a quote from Wikipedia concerning water vapor:
“Water vapor is a greenhouse gas in the Earth’s atmosphere, responsible for 70% of the known absorption of incoming sunlight…”
So – increased amounts of water vapor in the atmosphere simply must reduce the amount of energy that reaches the surface – and in turn the amount of IR back radiation from the surface (since there is a smaller total amount of energy to reflect).
This would also be hard to detect by sattelite, since there would still be an imbalance in/out, but the surplus energy will never reach the surface. Measurements will indeed show an increased imbalance between incoming and outgoing energy, but the energy will actually be absorbed further up in the atmosphere by the increased amounts of water vapor and it will never show up as IR back radiation and thus never be absorbed by CO2 or CH4…
Sure – the atmospheric temp. will be higher than it was before if water vapor increases, but CO2, CH4 and any other surplus anthropogenic gases won’t play any special role due to their IR absorbing capabilities.
This would be a true negative feedback, which can never be measured by sattelites that simply measures the in/out energy budget.
Please excuse the choppy language, I’m neihter a scientist nor a native speaker of the English language.
Patrik, You have a pretty severe misunderstanding of radiative balance. First, the vast majority of the sun’s energy is in the visible. Second, even if greenhouse gasses absorb incoming IR, that energy is STILL absorbed and so warms the planet. Third, the only way that energy would not warm the planet would be if it were re-radiated to space, in which case it would be visible to satellites and have the spectrum characteristic of the temperature of the atmospheric layer that radiated it.
Now this brings up another question: If you are not a scientist, then why do you think you understand science better than the scientists?
Hint: You don’t.
Re 213 Patrik
Water vapor is responsible for a majority of the absorption of solar energy *within the atmosphere*, which itself is less than half of the absorption of solar energy by the Earth (over half of solar heating is at/within the surface material).
Besides that:
Consider the approximation that convection is completely zero above the tropopause and that convection will not with a lapse rate smaller than a convective lapse rate and that convection easily keeps the lapse rate from getting any larger than the convective lapse rate. … (to be continued)
There is just never going to be 100% certainty about any future prediction, especially when the thing being predicted is complex and when the time involved begins to stretch out. It’s just not possible. But that is what the public demands. Thing is, with as many economic implications as GHG emissions mediation has that demand is not unreasonable.
When you factor in widespread rightwing suspicion of things scientific, which suspicion their talking heads regularly feed into, together with some good historical reasons for a general skepticism (other “scientific truths” have fallen by the wayside over time) the task of convincing people of the need to protect our planet from AGW gets downright difficult. Factor in an active corporate disinfomation campaign and you’re fighting a real uphill battle.
This is probably a case where convincing the public 100% simply cannot be achieved so it’s likely be a waste of time to try. Many people have become so jaded that only an in their face emergency will do it. By then of course it’s too late.
That’s why I prefer the most concrete evidence I can see, photographic evidence.
http://doublexposure.net/photos.html
http://nsidc.org/cgi-bin/glacier_photos/glacier_photo_search.pl?collection=repeat
http://www.nrmsc.usgs.gov/files/norock/repeatphoto/Pairs/RepeatPhoto_pairs_Fullset_compr.pdf
http://www.swisseduc.ch/glaciers/big_melt/index-en.html
http://www.extremeicesurvey.org/index.php/galleries/
Still even with this I am not 100% certain of what the future holds myself. I happen to see AGW as just one of many important environmental threats, so I get edgy at predictions. There’s alway a random factor in any situation, and we are learning new things all the time. I think greedy corporatists love it when “the intelligentsia” make predictions because they so often fail. Then these corporatists can then tie that failure to other environmental issues in an effort to get the public to simply dismiss it them all, thus giving them more time to rape and pillage our planet.
(cont. from my 215)…
In that case, outside the convecting layer, equilibrium is achieved by have a temperature distribution such that net LW cooling balances net SW (solar) heating. Above the tropopause, at any level, the net downward SW flux (equal to the amount of solar heating below) equals the net upward LW flux. Below the tropopause, it is the sum of upward non-radiative fluxes and net upward LW flux that balances the net downward SW flux. In order to sustain thermally-direct convection (setting aside thermally-indirect motions that might extend into the ocean or upper atmosphere, driven by kinetic energy produced within the troposphere), there must be some net radiative (SW+LW) heating lower in the troposphere and some net radiative cooling higher in the troposphere (higher and lower being relative; it can be the case that all the heating is at the base of the troposphere (surface) and the cooling is distributed over the rest of the troposphere).
The net heating or cooling of a layer is equal to the difference in net fluxes at the base and at the top of the layer.
The reason for the existence of the (radiatively-forced) troposphere, when one is sustained (as on Earth or Venus or…) is that, in order to eliminate any net radiative heating or cooling, for the given optical properties, the lapse rate becomes larger than what convection would sustain within some layer – thus being (conditionally or absolutely) unstable to convection. Convection would then occur, cooling the lower portion of that layer and heating the upper portion. Note that in doing this, it tends to increase the lapse rate at the boundaries (for example, at the top, there is convective heating below and lack of convective heating above), so that when radiative-convective equilibrium is reached, the convecting layer may extend beyond the original layer that was larger than the tropospheric lapse rate. (Also, it’s possible for kinetic energy to drive thermally indirect motion that can actually pull heat downward, with this process extending outside the region where convection is radiatively-forced, but we can set that aside for now.)
Within this troposphere, temperature at one level is related to temperature at another as determined by the tropospheric lapse rate; however, the temperature of the layer as a whole (using the temperature at some reference level) (when the troposphere is heated via convection/conduction/diffusion from an underlying surface, this ‘whole’ includes that surface) must be such that, for the given optical properties, the radiative fluxes…
(setting aside geothermal and tidal heating, which are relatively tiny for planets like Earth, Venus, Mars, Mercury, and the Earth’s moon, etc. – planets like Jupiter are a different matter (and Io?))
into and out of the whole layer must balance. Thus, when the convection extends (via conduction/diffusion) to the surface, the temperature at any reference level of the surface+troposphere, given the tropospheric lapse rate, and optical properties, must be such that the net upward LW flux at the tropopause is equal to the net downward SW flux at the tropopause – note that this includes solar heating both at the surface and within the atmosphere. Unless the solar heating is redistributed so much that the troposphere lifts off the surface, the temperatures beneath the tropopause are (in this approximation) unaffected by lifting some solar heating off the surface and redistributing it through the troposphere.
Hence the importance of tropopause level forcing.
If the solar heating within the surface+troposphere could be redistributed upward so that the solar heating of the surface were less than the net LW cooling of the surface, then the troposphere could lift off the surface with a reduced lapse rate below (the temperature would fall at the surface to bring the temperature into radiative equilibrium, though that would still depend on the temperature of the air above at various levels, depending on optical properties. But I wouldn’t expect an increase in water vapor to do that, because as water vapor is increased, it becomes nearly completely opaque to almost all LW radiation when it still lets a lot of solar radiation through (this large opacity is limited to lower levels of the troposphere and thus leaves room for CO2 and clouds, etc, to change the net LW flux at the tropopause and above; this is because the water vapor mixing ratio generally drops ‘precipitously’ (pun intended) with height within the troposphere. There is also less line broadenning and (I think) decreased line strength with increasing height, but that affects all gases (though maybe not equally?)). Thus, at the point where the net downward SW flux is very small, the net upward LW flux may still be much smaller, necessitating some non-radiative heat flux; only if (at/near the surface) that non-radiative heat flux were so small that it could be carried by conduction/diffusion with a lapse rate smaller than the tropospheric lapse rate (and that would have to be very very small) would the troposphere disconnect from the surface.
(As it is, LW cooling of the ocean is concentrated within a thin surface layer, while some wavelengths of SW heating penetrate tens of meters or more (depending on turbidity), and that can drive convection within the upper layer of the ocean. Also, evaporative cooling (both via effect on temperature and salinity) at the surface can drive mixing or overturning. Freshenning of the surface water by precipitation or other sources can inhibit overturning, while winds (and tides and planckton) can supply kinetic energy to force mixing against a tendency to become stably stratified. Of course, when water is sufficiently near the freezing point and sufficiently fresh, thermal expansion is negative, and spontaneous convection would be driven by cooling from below or heating from above; a sufficiently deep portion of a freshwater lake may tend to be near 4 deg C while the upper portion gets warmer in summer and freezes in winter.)
—
Keeping the total solar heating constant, an increase in solar heating above the tropopause would have a negative radiative forcing at the tropopause level by shading it. With stratospheric adjustment, the forcing could be smaller because the stratosphere would warm to increase the LW fluxes out to balance the increase in SW heating, and some of that change could be for the downward LW flux at the tropopause (a decrease in downward LW flux at the tropopause can occur for stratospheric cooling caused by GHGs – I say ‘can’ because it depends on the optical thickness of the stratosphere at the wavelengths where it can emit LW radiation, and if the optical thicknesses are high, it depends on where the warming or cooling occurs within the stratosphere. On Earth, the stratosphere is not very optically thick over most of the spectrum where it is significant (this being mainly from water vapor absorption bands in the stratosphere), so it radiates about the same flux upward to space as it does down at the tropopause even though the temperature is generally warmer in the upper stratosphere.)
However, an increase in absorption of solar radiation in the stratosphere or troposphere can reduce the albedo (by absorbing photons that would have been or have been reflected) and increase the total solar heating.
What about reducing the feedback section and adding climate sensitivity, which is the sum of all feedbacks? This would define and counter a skeptic argument. I still think answers to skeptic arguments is a good use of space.
Probably the best that can be said is:
This is the evidence we have to date.
This is what we believe that evidence means, and this is why we think that. However we acknowledge that there can never be 100% certainty in future predictions. Thus there is a chance that we are wrong; we estimate that our level of certainty is this while our level of uncertainty is this.
Taking all of this into account this is what we believe will likely occur we fail to act on GHG.
We believe that based on those risks the right thing to do would be to do something concrete about the issue.
This is what we propose to be done about the issue, and this is what we estimate the cost would be to do it.
===
An idea, new reports are coming in all the time and the public wonders how they relate to the overall picture. Perhaps wwe should have a system where every new legitimate study (by legitimate I am leaving out conflict of interest studies by industry hacks. Those studies should first be reproduced by others before inclusion) can be factored into a certainty/uncertainty index for quick checking by the public, e.g. a particular report now increases the certainty factor from 95.23% to 95.25% or perhaps lowers it from, 95.25% to 95.23% …
209: Lubos is right about that (maybe only about that). Energy is conserved in nuclear processes. All that E=mc^2 tells you is that a massive object at rest has an energy equal to its mass (in appropriate units). I think the post really should be corrected on this point; it’s a shame to give ammunition to right-wing zealots by getting a basic physics point wrong in a discussion like this (it’s a physics point that has essentially nothing to do with the argument, of course, but it’s still silly to get it wrong).
I raised a genuine objection at 184 and it has been ignored. At 188 Ray Ladbury talks about IR radiation, but my objection is that HEAT loss occurs at night, and the “simple recipe for GHE” explanation seems to assume permanent pervasive high noon for each point of the globe. Perhaps it is the extreme simplification that rasmus has done – and for which I am grateful. Nonetheless I would also be grateful if we could ask whether latitude, longitude, the earth’s daily rotation and its yearly circumnavigation of the sun could also be remembered. If the Earth loses its heat at night, and if the night lasts half a day at the equator, and half a year at the poles, then why is the night not long enough to lose the extra heat that comes when C02 goes up by a tiny fraction? If C02 goes up by a few parts per million then why does not the speed of heat loss at night slow down by a similar proportion?
Re 220 onymous – Energy is conserved if mass is considered a form of energy; outside of nuclear reactions and relativity, mass is often not considered energy, so I think it might be a matter of perspective (?)
Please not that there is nothing magic about molecules having three atoms to have vibrational and rotational absorption. Homonuclear diatomics have no change in dipole moment with vibration, so there is no interaction with dipole radiation without changing electronic state, which is too much energy for IR. The same is not true of hetero-nuclear diatomic molecules.
See http://how-it-looks.blogspot.com/2010/02/molecules-and-radiation-iii-vibration.html
Can we call optical thickness per unit distance (or per unit mass path or whatever other coordinate is convenient) ‘optical density’? If we could, ‘optical density’ = cross section per unit volume (cross sections can be absorption, scattering, extinction, or emission; emission = absorption at least for emission in one direction and absorption from that direction, at LTE, at a particular frequency, and when it matters, at a particular polarization; extinction cross section = scattering cross section + absorption cross section).
About stratospheric cooling:
Increasing LW optical thickness (emphasizing absorption/emission, as opposed to scattering) does this:
Case 1 – constant optical thickness over all LW frequencies:
1a. – isothermal stratosphere, positive tropospheric lapse rate: lapse rate :
The upward LW flux at the tropopause (first, by blocking radiation from the surface and replacing it with less radiation from a cooler troposphere, then, by blocking radiation from the warmer lower troposphere and replacing it with less radiation from a cooler upper troposphere, etc.) – this saturates when all the radiation at the tropopause is emitted from very near the tropopause.
The downward LW flux increases (by replacing the darkness of space with radiation emitted from the stratosphere) – this saturates when the stratosphere hides almost all of the darkness of space.
Both of these effects reduce the net upward LW flux at the tropopause.
When both of those effects are saturated, the net LW flux at the tropopause is zero (that tends not to be sustained in equilibrium climate because there has to be some net LW flux out to balance the net SW flux in – the climate response may involve a shift in the tropopause to ‘unsaturate’ the LW flux (such as by shifting upward to make the stratosphere thinner)).
The upward LW flux at the top of the atmosphere declines; this saturates when all fhe flux is coming from the isothermal stratosphere.
If the decrease in net upward LW flux at the tropopause is greater than the decrease in upward LW flux at the top of the atmosphere (which it will be, because the LW fluxes emitted from the stratosphere up and down both increase, and the decrease in the upward LW flux at the tropopause will be greater than the decrease in the upward LW flux into space that originated at/below the tropopause because of stratospheric absorption), the difference is a LW cooling of the stratosphere; holding the troposphere and tropopause steady, the stratosphere will cool until the downward LW flux at the tropopause and the upward LW flux to space have together decreased to bring the stratosphere back to equilibrium; this reduces the change to the net upward LW flux at the tropopause. When the troposphere and surface warm up to bring the net upward tropopause LW flux back into balance (equaling the net downward SW flux at that level), this increases the LW flux up into the stratosphere, and because some of that is absorbed in the stratosphere, the stratosphere warms a little bit and so there is a feedback in the downward LW flux at the tropopause causing some more warming below. The stratosphere could return to it’s original temperature – this will at least be true in the portion that acts like a skin layer if there is no albedo feedback and if there is no direct solar heating of the stratosphere (in which case, in order to be isothermal, the whole stratosphere must be a skin layer). Note that any solar forcing or decrease in albedo will tend to increase the stratospheric temperature even if there is no direct solar heating of the stratosphere, because the stratospheric temperature is a function of the upward LW flux from below. If there is direct solar heating of the stratosphere, then the full equilibrium involves a stratospheric cooling; as the LW optical thickness increases, the layer that is thin enough to be a skin layer shrinks, and by shrinking (while holding the distribution of solar heating constant), reduces the solar heating of that layer, so that the skin layer’s temperature declines towards the value it would have in the absence of direct solar heating.
1b. The stratosphere’s temperature increases with height (due to direct solar heating), either from the base or above a lower isothermal portion.
In that case, if the cooler lower isothermal portion is thick enough relative to the whole stratosphere, the downward LW flux will increase until it saturates; otherwise, it might increase, and then decrease as the lower cooler stratosphere blocks radiation from the warmer upper part; it would saturate when all the downward radiation at the tropopause originates from an isothermal portion or else from very near the tropopause.
The upward LW flux to space will decrease until a sufficient portion originates in the upper stratopshere; it will then start to increase.
If the lower cooler portion of the stratosphere is sufficiently thin and the upper warmer portion is isothermal over some thickness at the top, then when the optical thickness is large enough, farther increases could reduce the LW fluxes out of the stratopshere (by reducing the downward flux at the tropopause while the flux to space is saturated). If this effect overcomes the cooling from the portion of the reduced upward LW flux from below that would have been absorbed, then the stratosphere as a whole could warm. However, that warming would not be evenly distributed; There will be some cooling in the uppermost stratosphere at least because the LW emission to space gets concentrated toward the top of the atmosphere. As LW opacity increases, the top of the atmosphere could eventually approach the skin temperature as it would be with no direct solar heating.
—-
(Note that if there is a very very small amount of direct solar heating in a very very thin layer at the top of the atmosphere, it would be possible to reach the skin temperature (for no direct solar heating) in a portion of the uppermost atmosphere below the solar-heated top layer, if the LW opacity is not too large.)
——-
Case 2. optical properties vary with wavelength; optical thickness is increased at some wavelengths and not others.
The LW fluxes change at each wavelength where optical thickness is added, in the same way (qualitatively) that they do for all wavelengths in case 1.
However, the LW fluxes respond to temperature at all LW wavelengths. But it is true that (absent other feedbacks) the temperature changes so that, at the tropopause and above, the net LW flux leaving a layer in total changes to balance the optically-forced changes in the net LW flux leaving that layer.
Stratospheric cooling would reduce the downward LW flux at the tropopause at wavelengths where the stratopshere has some optical thickness (but this is not necessarily the case if the optical thickness is large). It would also reduce the LW flux to space at wavelengths with some optical thickness within the stratosphere. But at wavelengths where the stratosphere is optically thick, the change in the LW flux depends more on the stratospheric cooling in the closest portion of the stratosphere; the base of the stratosphere could actually be warming, in which case, the downward LW flux from the stratosphere would actually increase at those wavelengths where the optical thickness of the stratosphere is large enough.
When the troposphere+surface warm, the upward LW flux increases at all wavelengths (except, see next paragraph). Some of that would be absorbed by the stratosphere, causing warming of the stratosphere (not necessarily evenly distributed; it would tend to be concentrated in the base of the stratosphere at wavelengths with larger optical thickness, assuming some of the increased upward LW flux occurs at those wavelengths (see below)).
However, the top of the troposphere and the base of the stratosphere have the same temperature (the tropopause temperature), so the changes in dowward LW flux and upward LW flux at the tropopause must be equal and thus have no effect on the net LW flux at those wavelengths where the optical thickness is so large that both fluxes originate from isothermal regions or very near the tropopause. Also, the tropopause will tend to shift a bit, and if the tropopause temperature manages to stay constant while the troposphere and surface warm (by rising in proportion to the warming divided by the lapse rate), then the resulting change in downward and upward LW fluxes will both individually be zero at wavelengths where there is sufficiently large optical thickness.
Thus, after all levels have together reached a new equilibrium, even assuming no SW (albedo) feedbacks, there could be the same upward LW flux at the tropopause in total, or there could be slightly more if the tropopause shifted across a layer that absorbs solar radiation (thus ‘sequestering’ solar heating into the troposphere from what was the stratosphere), but the amount of that flux that is absorbed by the stratosphere could decrease because it might be shifted into wavelengths where the stratosphere doesn’t absorb as much of it. That would tend to cause stratospheric cooling (somewhere within the stratopshere).
…”That would tend to cause stratospheric cooling (somewhere within the stratopshere).” – even if the stratosphere had no direct heating from the sun.
PS about feedback from shifting the tropopause:
The only effect on the stratosphere would come from the changes in the upward LW flux at each level within the stratosphere caused by warming of the layer that was above the tropopause that is now below, because while the total solar heating and the total LW optical thickness of the stratophere may be reduced, the shift in the tropopause doesn’t directly affect either for the portion of the stratosphere above the new tropopause.
But the upward shift in the tropopause could reduce the downward LW flux at the tropopause by decreasing the LW optical thickness of the portion of the atmosphere remaining above. However, by bringing the warmth underneath the tropopause closer to the rest of the remaining stratosphere, the remaining stratosphere could warm up a bit and radiate a greater LW flux downward; if the LW optical thickness were only either large or insignificant at any wavelength, then (would they tend to balance resulting in no net change in downward LW flux from that effect?)
Confused, Earth’s surface radiates day and night. As such it loses energy day and night. The only difference time of day or time of year or longitude or latitude make is the amount of INCOMING radiation. The processes are the same, and the simple picture Rasmus defined applies. In point of fact, the thing to remember is that Earth absorbs energy over 2pi steradians and radiates over 4pi steradians. Does that make it any clearer?
Patrick 027 says:
This distraction provides support for deleting the reference to it in the lead article. It was supposed to be about the greenhouse gas mechanism! Before you can say Jack Robinson, we’ll get on to the other populist nonsense attributing nuclear weapons to poor old Einstein in 1905.
You could go further and argue that heat can never be created, outside of certain dissipative mechanisms ….that too is all a matter of perspective. I shall not reply again to this OT stuff.
—————–
This is on topic. I don’t agree with your dismissal of my question about pressure broadening, which includes temperature. It is obviously vital otherwise the spectral absorption would consist of a series of delta functions. The question is whether we can get simplify it so as to avoid getting bogged down. One way would be to ask a climate model.
A simple question:
Suppose GHG stabilize at some level, say double the pre-industrial one.
Will the forcing, and temperature rise, continue or will they stabilize too, at some (higher) level ?
Is the danger of a runaway positive feedback just a vague fear, or is it based on robust calculations ?
I think there should be a note at least that usually the term ‘enhanced greenhouse effect’ means that portion of the greenhouse effect which is owing to human activity. In the article, it might be owing to some natural fluctuation.
Re: my previous comment
Not delta functions but sharp spikes. (I may make more mistakes then George Monbiot but I aim to correct them a bit quicker.. see Guardian)
222 Patrick 027 wrote: “Energy is conserved if mass is considered a form of energy; outside of nuclear reactions and relativity, mass is often not considered energy, so I think it might be a matter of perspective ”
No. You can directly estimate the energy released in a fission event without reference to E=mc^2. The theory of nuclear fission is nonrelativistic. The mass lost in E=mc^2 is tiny compared to the total mass, not as tiny as the mass lost in a chemical reaction, but tiny none-the-less. The argument that was used in the 30s to estimate the energy released in a fission event follows. It is identical to the argument one would use to estimate the energy released in a chemical reaction. No change in mass is assumed.
For a nucleus with Z protons and radius R the binding energy, E = Z^2 e^2/R where e is the charge on a single proton. They first estimated that E is about 10^8 times what it would be for a low Z chemical reaction because for, say, uranium, Z~=100 (Z=92 but we’re among friends). R is about 10^4 times smaller than atomic distance scales. Thus the binding energy in a nucleus is 10^8 times a typical chemical bond. It turns out that there is an empirical relationship for R based on the number of nucleons in a nucleus: R = ro A^1/3 where A is the number of neutrons + number of protons in the nucleus. If we split the nucleus into two identical pieces so that A-> A/2, Z->Z/2, R-> R / 2^1/3 ~= R/1.25 .
The binding energy of the two remaining pieces is: 2 x (Z/2)^2 1.25 E = .6 E
The first “2” comes from there being 2 pieces. Thus the energy released from the fission event is about E-.6E = .4 E and remember that E is about 10^8 times the energy released in a typical chemical reaction.
There are numerous points in this article that need work and deleting the remark about conservation of energy is a really easy one. The stuff on optical thickness needs improvement too.
Perhaps a bit nitpicky, but you imply that most or all of the energy in solar radiation is visible. This is not correct – only approximately 43% is visible [http://www.ucar.edu/learn/1_3_1.htm] . The important point is that most of the non-visible is “near” infrared which is much less absorbed by GHG molecules than “far” infrared.
Ray, I think your making Confused’s explanation too complex — or I’m missing his question. Climate assessments are all based on long term averages, both time and location. Incoming solar is normalized and assumed mathematically to enter evenly throughout the 4pi steradians of the whole globe, 24 hours per day, 365 days per year. The fact that it physically actually varies by (vertical) hemisphere, by time of day, by one day’s weather, etc. matters not a twit in the analysis. The fact that upwelling IR radiation actually is relatively constant over time and place is just conveniently makes it easier to plug into the analysis. One other point I think Confused was asking about: the radiation leaving and cooling the surface is not the same as that leaving and cooling the entire biosphere, nor does it include the downwelling IR radiation returning to the surface.
Jacob #228 : I think that your question sadly illustrates how it is difficult to explain properly simple things. The answer should be obvious to anyone who is interested in the GW issue, and still more after having read a supposed “simple presentation” for general audience. The forcing IS directly linked to the increase of GHG so it will of course stabilize if GHG concentration stabilize. Concerning the temperature , it is very important to realize that GHG are not a heater , but a blanket. They do not produce heat, they hinder the heat transfer throughout the atmosphere. So the temperature increases only transiently, because of two phenomena
* the Earth takes some time to reach its equilibrium temperature (there is a relaxation time)
* the GHG concentration is slowly increasing so the Earth cannot yet stabilize (there is also a growth time)
BUT in the case of a stabilization of GHG concentrations it should be obvious to anyone that the temperature would also stabilize after some relaxation times.
It is also important to understand that the main reason for the uncertainty in climate sensitivity (in my opinion) is that the observed slope is a COMBINATION of both time scales, but it is very difficult to determine the influence of each one. The same slope could be observed with a large sensitivity AND a long equilibration timescale, or a small sensitivity and a small time – but the final result would be different, since in the first case the temperature would keep growing during a longer time and reach a higher asymptotic level. The problem of sea level rise is pretty much the same. This is rather uncomfortable since it allows many people to claim that “wait , the sensitivity could be much larger and the timescale be also much larger, so the final result would be much more catastrophic” , leading to all kind of dire predictions. This is not false — it COULD be higher – but the question is : how to compute properly this risk , to base a cost-benefit estimate ? this is really tricky. The principle of precaution would give absurd results if we should act as if the worse predictions were certain – nobody should go out of his home in the case of a meteorite could hit him for instance. Much of the complexity of the GW issue arises from this uncertainty, IMHO.
Here is a denialist critique re AGW (the “log function” argument) I saw on a blog. Any good answers?
[blockquote]”The radiative forcing is a log function of CO2 concentration.”…This is an example of how the warmers outsmart themselves. A linear CO2-temp correlation is actually inconsistent with greenhouse warming, not evidence for it. Given this inconsistency there are 3 generic possibilities. First, the correlation is a coincidence. Second, there is an unknown inverse log positive feedback mechanism that magically makes up for the log decline in CO2 forcing with concentration level. Third, the causality runs the other way and the CO2 increase is due to the temperature rise, as some have argued and the ice core data seems to show.[/blockquote]
I know it is a log function relationship, and I pointed out that for short time frames it looks more or less linear, but I am not sure about how long that timeframe is (when the relationship between CO2 & warming can be viewed as linear. Is it a century, or what?
Also, if anyone has a good response, I’ll post it on that Catholic blog.
How about this as a [reason](http://www.guardian.co.uk/commentisfree/video/2010/jul/08/david-mitchells-soap-box-climate-change to act on climate change instead.
Re 177 McGahill – another way of putting that is that GHGs help the surface, or surface plus some portion of the atmosphere (when optical thicknesses are large enough) retain heat in the same way that a thermos bottle slows the cooling of a warm liquid inside. As opposed to the way that the heat capacity of the liquid itself would slow it’s temperature change.
Re 234 Lynn Vincentnathan – a few helpful points
1. Any continuous smooth relationship can be approximated as linear over a sufficiently small interval. It’s conceivable that the small changes in CO2 that the blog was referencing were too small to show the logarithmic nature of the relationship.
2. Of course, climate sensitivity needn’t be constant over temperature, though it is also true that greenhouse forcing itself depends on temperature – but see point 1.
3. I don’t think it was ever asserted that the temperature would increase linearly in proportion to CO2.
234: Lynn said about logs and time “I pointed out that for short time frames it looks more or less linear, but I am not sure about how long that timeframe is (when the relationship between CO2 & warming can be viewed as linear. Is it a century, or what?”
I see a bit of confusion in this question. The relationship for climate sensitivity is based on the concentration of CO2 and as far as I know time doesn’t enter into it. If earth’s co2 concentration (which I’ll just write as co2 to avoid having to say “concentration” all the time) is doubled then the mean temperature change is described by a function which is approximately logarithmic. Thus one can write:
dT = c log(co2/co2_0) where co2_0 is the reference concentration. If the temperature change, dT, is 3K per doubling then c= 3K/log(2) ~ = 4.3 K
As far as I know, this is basically a result from a lot of climate simulations. I don’t think there is any simple first principles derivation of the logarithmic dependence. You will occasionally hear people claim that Beer’s law is somehow responsible for the logarithmic dependence but this is nonsense. It doesn’t hold on venus for example.
If you were to double the concentration overnight and then wait for the new mean temperature to settle down it would take some time. First the atmosphere and top couple meters of ocean would equilibrate which takes about a year. Next the top few hundred meters of ocean would equilibrate taking about 30 years and then the remaining part of the ocean would take about 1,000 years to equilibrate. At the end of the 1,000 years radiative balance will once again be restored and the mean temperature would have increased by about 3K. But it takes time. My picture is admittedly crude; there are all sorts of upwellings and downwellings of heat into and out of the oceans so the atmospheric temerature goes up and down, but my timescales are about right. Note that the logarithmic dependence doesn’t specify the time course of the increase in temperature. It just says that if double CO2 and wait long enough the temperature will go up by about 3K. Since the atmosphere heats before the deep ocean we see the temperature increase fairly fast (compared to the deep ocean bottom). If we lived in Atlantis we probably wouldn’t have noticed it yet unless we had very accurate ways to measure the temperature in the deep ocean.
About smoothness etc: for |x| << 1 it turns out that log(1+x) ~ = x. Thus for small changes in co2 = co2_0 + dco2 where dco2 << co2_0 we can use the property of the log mentioned in the preceding sentence to write:
dT ~= c dco2/co2_0
which does show a linear temperature change for small dco2/co2_0 . I hope this helps a bit.
Re 227 Geoff Wexler
This is on topic. I don’t agree with your dismissal of my question about pressure broadening, which includes temperature. It is obviously vital otherwise the spectral absorption would consist of a series of delta functions. The question is whether we can get simplify it so as to avoid getting bogged down. One way would be to ask a climate model.
I didn’t intend my 165 to be a dismissal. I agree it is important (see my 177 https://www.realclimate.org/index.php/archives/2010/07/a-simple-recipe-for-ghe/comment-page-4/#comment-180079). I think the justification for main post refering to broadenning (and would have presumably refered to line strenth’s temperature dependence) as being secondary was that, while important in total and also to variation over altitude (via T,p) in optical properties per unit material , they can also be feedbacks, but as climate feedbacks, they are relatively minor compared to the Planck response and some other important feedbacks like water vapor and albedo.
Maybe one way to simplify it would be to say that the optical thickness added by some amount of material depends on temperature, pressure, and composition, but that, for climate changes that are not too large, the optical thickness per unit material can be approximated as constant at a given pressure level in the atmosphere.
Or more briefly, optical thickness varies in proportion to the amount of a substance that supplies it, but also depends on the conditions in which that substance is found, but that we can set that dependence on conditions aside for introductory purposes.
The analysis is false.
It says the only source of incoming energy is solar insolation.
Are you denying that the force of gravity adds energy by the sun & Moon causing tidal effects and hence friction energy?
Are you denying that Gravitational potential energy, when the Earth moves closer to the sun or to another planet will result in potential energy being reduced and becoming more kinetic energy as measured by temperature? ie it warms
Then are you saying that simply adding more GHGs will cause more warming, even when all 1366 W.m^2 is already in use to create the existing 32c of GHE, but adding more GHGs will result in more than the 1366 being used to create more GHE? IF this is true then why doesn’t more warming result when the amount of GHGs water vapor increases from 33% to 100% when it rains?
Arrheius’s 1896 thought experiment said that when you add a GHG to a supply of photons you get warming. So what happens when you add a GHG, but all the photons are already in use creating the existing 32C of GHE? Do you not get an excess of GHGs and NO ADDITIONAL WARMING, because there is no energy available?
Sorry but you are mis applying the real greenhouse effect.
RE #238, Thanks, John, but it is still a bit confusing. You say that if we double CO2 and wait long enough the temperature will go up by about 3K. I’ve heard that is the most likely outcome. But I guess if it’s a log relationships, then it matters where you start when you double the CO2. Like if there are 800 ppm in the atmosphere and we double that to 1600, would it again increase by 3K? I’m assuming that they are talking about doubling CO2 from preindustrial 270ppm to 540 ppm or something like that, and it would be a different factor (not 3K) if we start way higher in ppm or way lower.
This may be new denialist argument — that the “warmers” are asserting a linear relationship, when in fact it is a log relationship — and everyone knows the log function goes pretty flat, so increasing CO2 is no real problem, as the “warmers” assume….
I suspect you should revise your discussion and graph of predicted vs observed surface temperature. We haven’t sent probes to measure the surface temperature of all planets and four moons. We do have measured temperatures for some places on our Moon (where the temperature varies about 200 degK between day and night) and Mars (where there are also large temperature swings), one datapoint for Venus (confirming that the surface is extremely hot) and possibly one for Titan.
Most of your “surface temperature” data is calculated from the spectrum of the radiation emitted by the planet (including a variable factor, emissivity). For the gas giants, we can’t directly observe radiation from the surface of the planet, so your “surface” temperatures presumably comes from some point high in the atmosphere. On your graph, planets and moons lie on the line when their “surface” temperature has been determined at a location where incoming and outgoing radiation are in equilibrium (adjusting the emissivity) and off the line when other locations are used. If the temperature of the atmosphere where incoming and outgoing radiation are in equilibrium had been used to determine the “surface” temperature of Venus, Venus would be colder than the Earth (because it has a much higher albedo)!
Dear Gavin, Chris,
When you would have taken the time to read what I have written, you would probably have reacted a bit more moderate. To start with, Gavin made the assumptions about the impact of possible latent heat loss to space, not I. Chris, you did no even bother to read what I posted. What I did post was my opinion, that there are numerous processes which are not commonly recognized in a “vacuum” that contribute to non radiative energy transfer. I guess most of the respondents have not realized that cubic space increases with the third power of the distance from the centre of the earth? This means that any molecule with an above average speed will travel a long way into space before colliding. But it will collide. And in the process it will cool down, transferring energy to outer space.
[Response: This is completely irrelevant for climate. – gavin]
Re 240 John Dodds The analysis is false.
It’s an approximate description.
It says the only source of incoming energy is solar insolation.
Which is a very good approximation for at least the inner planets (and their moons).
Are you denying that the force of gravity adds energy by the sun & Moon causing tidal effects and hence friction energy?
You’re forgetting the energy from combustion of fossil fuels and the much larger geothermal heat flux from the interior. But all these together are on the order of 0.1 W/m2 for Earth, and will be small for the other inner planets as well. They add heat to the climate system; whereever that heat is delivered to the climate system (at the surface, or mostly within the ocean for the tides), the effect is the same as if it were from the absorption of solar energy. So it would be like solar heatinger were on the order of roughly 0.04 % larger, with less than a tenth of that difference managing to penetrate deeper into the oceans than the rest. The effect would be negligible, and it wouldn’t change how the greenhouse effect works. (There is another affect of the tides – the forced motion (the viscous damping of that being the way that the tidal energy is realized as heat) – this has some importance to oceanic circulation but it does not have a big impact on global average surface temperature; of course processes in the interior of the Earth shape the geography and chemistry of the surface and atmosphere over time, but that’s really on a different level).
In other places in the solar system or elsewhere, where tidal and/or geothermal or some other heat source (like impactors or a stellar wind, etc.) is large enough relative to stellar heating to make a difference to the surface and atmospheric conditions of a planet or moon or asteroid, then these things could be taken into account with the known physics.
Are you denying that Gravitational potential energy, when the Earth moves closer to the sun or to another planet will result in potential energy being reduced and becoming more kinetic energy as measured by temperature? ie it warms
That’s not how it works. An object in the gravitational field of other objects has some gravitational potential energy and may have some kinetic energy. In a constant circular orbit about one other object, the two quantities stay constant; in an elliptical orbit or an orbit perturbed by other objects, there is some conversion between gravitational potential energy and the kinetic energy of the motion of the object through space. The only way such kinetic energy of these objects is converted to heat within or at the surface of such an object is via impacts with other objects (or via electric currents driven by each other’s magnetic fields, but in familiar interplanetary conditions, that’s not going to be significant either). On that note, there would be some (relativistic) doppler effect of the Earth’s motion on the energy of the photons it recieves from the sun (blueshift in morning, redshift at night, with the orbit and rotation being both prograde), but that’s probably insignificant (as are the gravitational lensing and redshift as photons reach, leave, or go by the Earth).
Then are you saying that simply adding more GHGs will cause more warming, even when all 1366 W.m^2 is already in use to create the existing 32c of GHE, but adding more GHGs will result in more than the 1366 being used to create more GHE? IF this is true then why doesn’t more warming result when the amount of GHGs water vapor increases from 33% to 100% when it rains?
Sorry but almost every part of that is wrong.
The ~ 1366 W/m2 solar flux at Earth must be divided by 4 and then multiplied by approximately 0.7 to get the global time average solar heating (because the Earth’s albedo for the solar radiation is 0.3 and, being nearly spherical, has a surface area 4 times it’s cross-sectional area).
Aside from (non-Planck response) feedbacks, that solar heating is not changed by the greenhouse effect or changes in the greenhouse effect. In climatic equilibrium, the same LW flux must go out as what comes in as solar radiation (plus the very small fluxes mentioned earlier). That flux depends on temperature and on the greenhouse effect. Changing the greenhouse effect changes where the LW fluxes come from within the system, and thus changes the temperatures required to have an equilibrium.
The concentration of some greenhouse gases and other agents (clouds), and the amount and vertical distribution of solar heating, and the temperature and it’s vertical distribution, all vary over the globe and over a year and over shorter-term internal variability. But none of that changes the basic physics and how it works. It just means that a complete description is more complicated. But in terms of global time averages, the requirement of balances in vertical fluxes still applies for climatic equilibrium.
Arrheius’s 1896 thought experiment said that when you add a GHG to a supply of photons you get warming. So what happens when you add a GHG, but all the photons are already in use creating the existing 32C of GHE? Do you not get an excess of GHGs and NO ADDITIONAL WARMING, because there is no energy available?
That sounds like Beer’s law. First of all, not all of the available photons are used for the present-day GHE, in the sense that some of the radiation from the surface does escape directly to space. Secondly, the phrasing ‘all the photons are already in use’ could mean a number of things – see above, this paragraph, below, whatever…
Thirdly, Beer’s law only applies when there is no emission of photons along the path considered. GHGs emit photons as well as absorb them, but they emit according to there temperature. In the most general description: because the atmosphere is generally heated by the surface, it must be colder than the surface, so that it absorbs more LW photons from the surface than it emits to the surface that are absorbed by the surface, and so that convection can carry heat from the surface to the atmosphere where there is a troposphere. The atmosphere reduces the outgoing LW flux to space by blocking radiation from below and replacing it with it’s own emitted flux, which, being colder than the surface, will be less than what it absorbed from the surface. There is a general decrease in temperature over height within the troposphere, so even if all radiation from the surface is blocked, the outgoing LW flux can still be decreased by having the colder upper troposphere block more radiation from the lower troposphere. What happens if there is no troposphere or what happens above the troposphere is different, but in that case, it doesn’t affect the surface temperature in the same way.
The paragraph immediately above the paragraph with the bold phrase should have been in italics indicating quotation.
PS had similar error in above response to 239 in response to 227 Geoff Wexler
Re 231 John E. Pearson – but isn’t the energy that comes from such reactions is equal to a loss of mass (at least, once the energy leaves (?)). But good point about chemical reactions (as in, it’s not just nuclear reactions).
Re 238 John E. Pearson (and/re Lynn Vincentnathan)
You will occasionally hear people claim that Beer’s law is somehow responsible for the logarithmic dependence but this is nonsense.
So true. At any particular frequency (wavelength), Beer’s law does allow and call for eventual saturation in some conditions, which would not be logarithmic but rather asymptotic, and would occur when, at the point considered, photons reaching that point are being emitted from places all at the same temperature as at the point considered. A logarithmic function doesn’t have an asymptote; it keeps increasing, just more slowly. The logarithmic proportionality of forcing to CO2 amount is due to the shape of the CO2’s absorption band (glossing over finer scale texture and some bumpiness, going away from the center of the band, optical thickness per unit mass path of CO2 decays exponentially, within certain limits) and that it’s central portion is saturated (at the tropopause level, the net LW flux is zero at the center of the band; at the top of the atmopshere, the upward LW flux has reached a minimum within the CO2 band and farther increases in CO2 increase the upward LW flux at the center of the band (at least before the temperature responds). Aside from the central rebound in outgoing LW radiation at TOA, the changes in LW fluxes caused by changes in CO2 occur at the edges of where the CO2 absorption occurs, and are proportional to an expansion of that interval of wavelengths, which is approximately the same for each doubling (until the amount of CO2 is so much that other parts of it’s absorption spectrum come into play).
John Dodds (81): Brilliant! Your gravity theory of climate change is somewhat similar to that of the unorthodox genius Steven Goddard. You should not even waste your time here – contact the Best Science Blog immediately!
Re 234 Lynn Vincentnathan
…the warmers …that Catholic blog
Maybe try recommending the movie “Evan Almighty” (it’s a good movie!)