What exactly is the greenhouse effect? And what does it look like if we view it from a new angle? Of course, we know the answer, and Raymond Pierrehumbert has written an excellent paper about it (Infrared radiation and planetary temperature). Computer code used in climate models contain all the details.
But is it possible to provide a simple description that is physically meaningful and more sophisticated than the ‘blanket around earth’ concept? I wanted a description that could be grasped by physicists. Without the clutter of too much details – just the essentials. A ‘back-of-the-envelope’ type derivation of the greenhouse effect.
The starting point was to look at the bulk – the average – heat radiation and the total energy flow. I searched the publications back in time, and found a paper on the greenhouse effect from 1931 by the American physicist Edward Olson Hulburt (1890-1982) that provided a nice description. The greenhouse effect involves more than just radiation. Convection also plays a crucial role.
How does the understanding from 1931 stand up in the modern times? I evaluated the old model with modern state-of-the-art data: reanalyses and satellite observations.
With an increased greenhouse effect, the optical depth increases. Hence, one would expect that earth’s heat loss (also known as the outgoing longwave radiation, OLR) becomes more diffuse and less similar to the temperature pattern at the surface.
An analysis of spatial correlation between heat radiation estimated for the surface temperatures and that at the top of the atmosphere suggests that the OLR has become more diffuse over time.
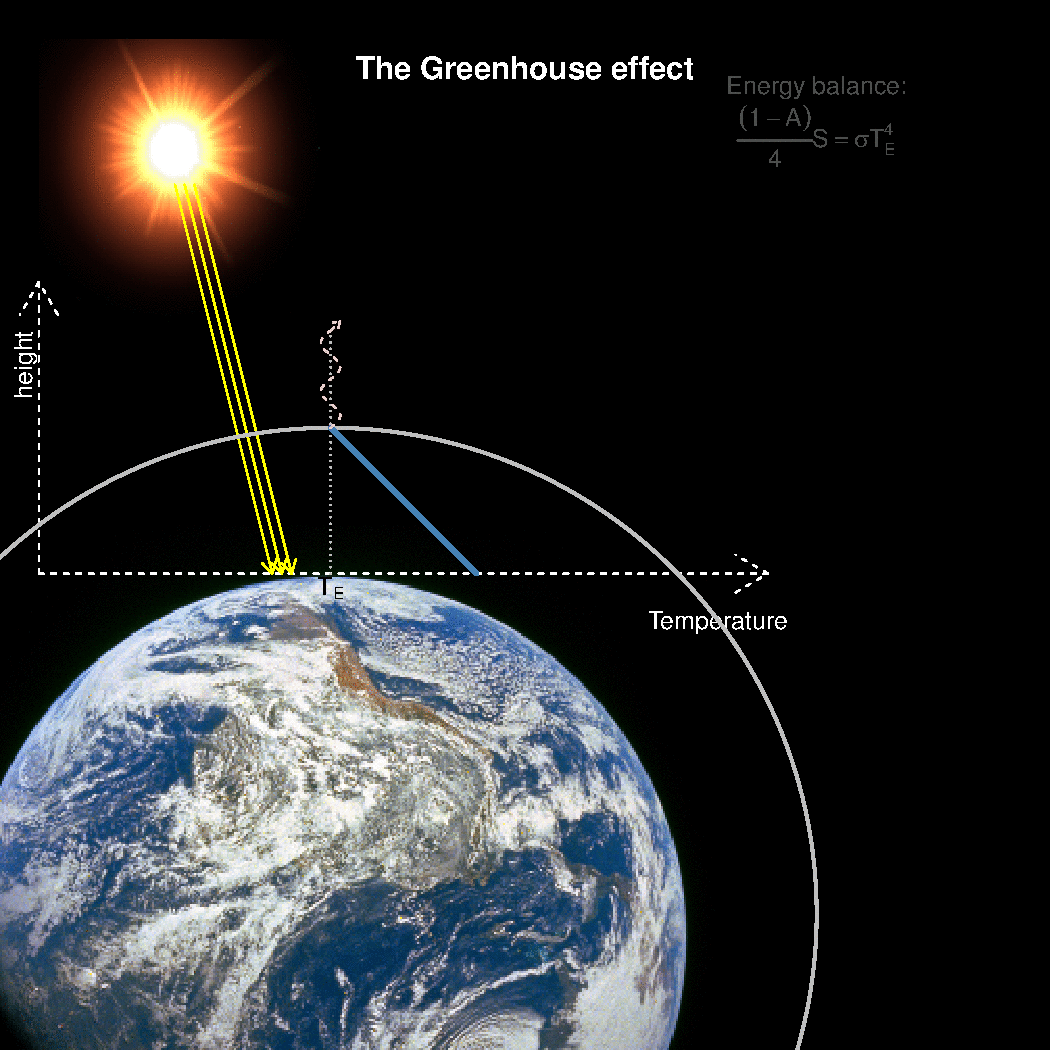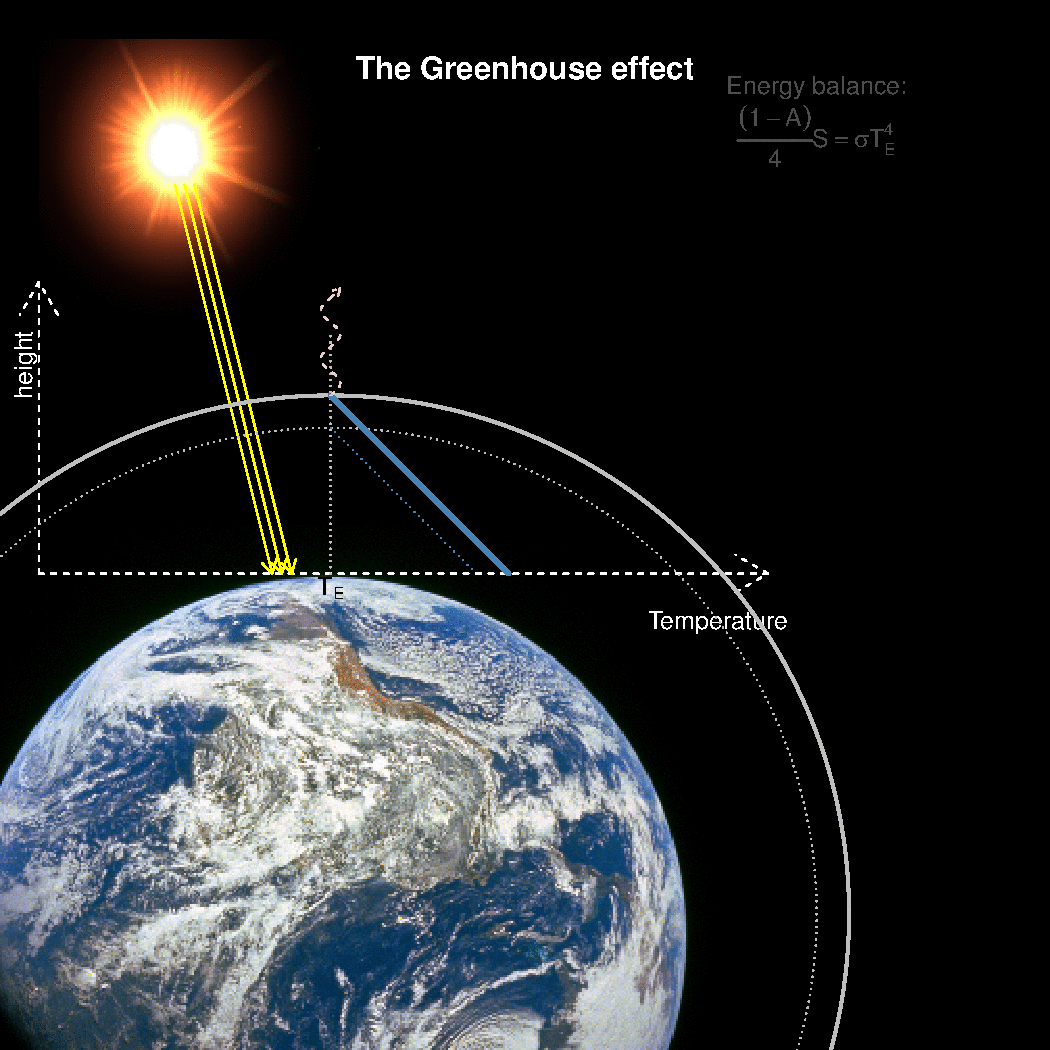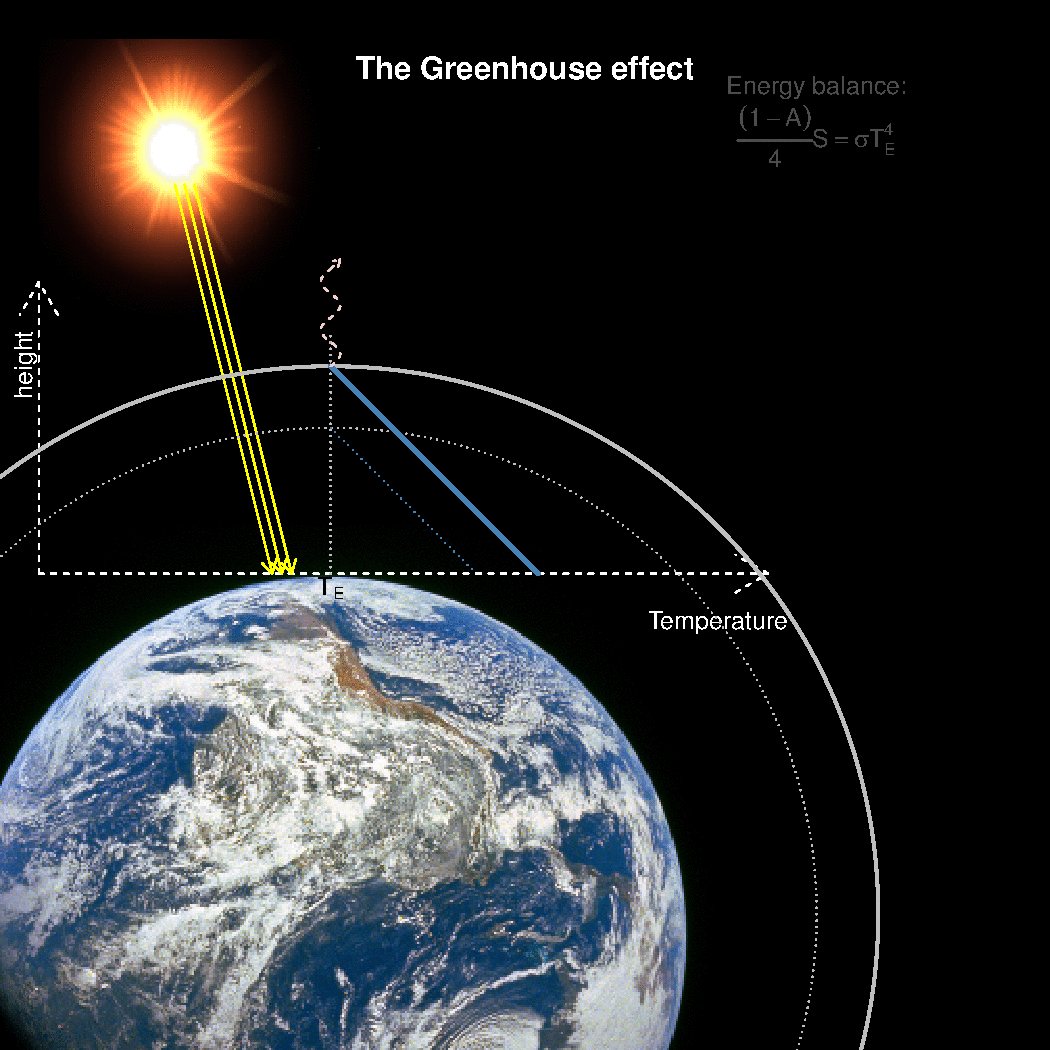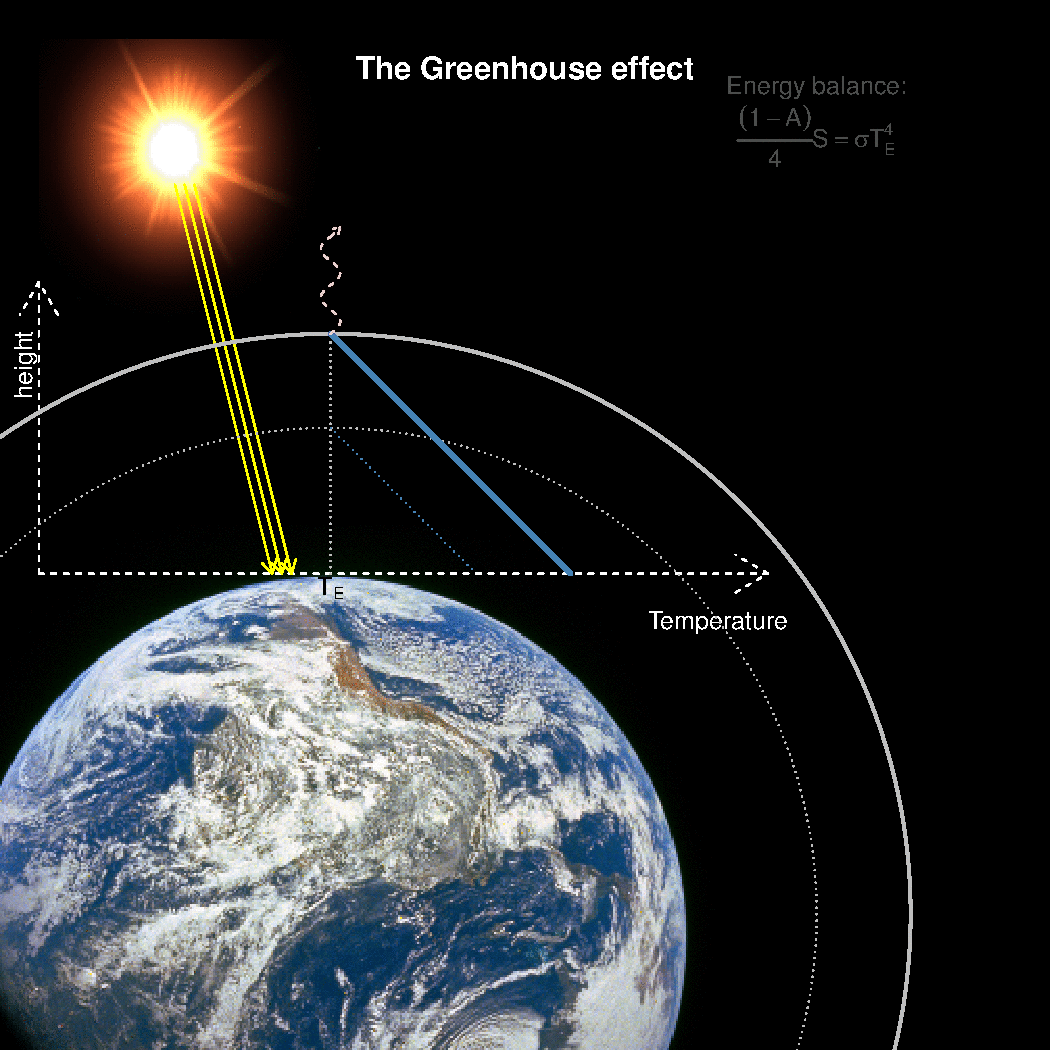
The depth in the atmosphere from which the earth’s heat loss to space takes place is often referred to as the emission height. For simplicity, we can assume that the emission height is where the temperature is 254K in order for the associated black body radiation to match the incoming flow of energy from the sun.
Additionally, as the infrared light which makes up the OLR is subject to more absorption with higher concentrations of greenhouse gases (Beer-Lambert’s law), the mean emission height for the OLR escaping out to space must increase as the atmosphere gets more opaque.
There has been an upward trend in the simple proxy for the emission height in the reanalyses. This trend seems to be consistent with the surface warming with the observed lapse rate (approximately -5K/km on a global scale). One caveat is, however, that trends in reanalyses may be misleading due to introduction of new observational instruments over time (Thorne & Vose, 2010).
Finally, the energy flow from the surface to the emission height must be the same as the total OLR emitted back to space, and if increased absorption inhibits the radiative flow between earth’s surface and the emission height, then it must be compensated by other means.
The energy flow is like the water in a river: it cannot just appear or disappear; it flows from place to place. In this case, the vertical energy flow is influenced by deep convection, which also plays a role in maintaining the lapse rate.
A popular picture of the greenhouse effect emphasises the radiation transfer but does not explicitly account for convection. As a result, it fails to explain the observed climate change.
Hulburt’s old model from 1931 included both radiative energy transfer and convection. It has now been validated against state-of-the-art data, and non-traditional diagnostics show a physically consistent picture.
An increased overturning can even explain a hypothetical slowdown in the global warming, and the association between these aspects can be interpreted as an entanglement between the greenhouse effect and the hydrological cycle, in which a reduced energy transfer associated with increased opacity is compensated by an acceleration of the hydrological cycle. This also makes a link with clouds.
The old conceptual model also explains why the so-called ‘saturation’ (which doesn’t exist on Venus) is a red herring, which is also explained in the report by the Copenhagen Diagnosis. I think those who present this argument have a poor understanding of what the greenhouse effect is all about.
A bold proposal: One way to view the greenhouse effect is the vertical distance between the place where incoming energy is deposited and where the average outgoing heat loss takes place. This distance depends on the concentration of greenhouse gases, and at what height the OLR can escape to space without being reabsorbed by air above.
The graphics below provides a crude illustration: the OLR is determined by Stephan-Boltzman’s law and the temperature at the same height, and the surface temperature is then given by the emission temperature, the emission height, and the lapse rate.
A more elaborate description is given in (Benestad, 2016), which was inspired by two posts here on RealClimate (here and here).
Not all of my colleagues may agree with my description of the greenhouse effect; it was a struggle to get this paper published. To my surprise, I realised that there are scholars with different ideas about it. However, I hope that my description will lead to more discussions and debate about the over-arching principles and our basic understanding of this phenomenon.
This also touches upon the question of climate sensitivity which is merely defined in terms of temperature change. A response to increased greenhouse gases could involve both a global warming and a speed-up of the hydrological cycle if the greenhouse effect and the hydrological cycle are intertwined. In other words, there could be more dramatic changes to the rainfall patterns than the temperature, but this doesn’t necessarily imply that the climate is less sensitive to the forcings.
References
- P.W. Thorne, and R.S. Vose, "Reanalyses Suitable for Characterizing Long-Term Trends", Bulletin of the American Meteorological Society, vol. 91, pp. 353-362, 2010. http://dx.doi.org/10.1175/2009BAMS2858.1
- R.E. Benestad, "A mental picture of the greenhouse effect", Theoretical and Applied Climatology, vol. 128, pp. 679-688, 2016. http://dx.doi.org/10.1007/s00704-016-1732-y
Jim Eager wrote in 44:
I am still finding it at:
http://www.globalwarmingart.com
#49 & related–
This is a bit pedantic, but I feel compelled to mention that actually, insolation is ‘majority IR’ by energy:
That’s from WIkipedia; the footnote clarifies:
But I have seen the same point made elsewhere. (No, I don’t recall where, exactly.)
Eyeballing the spectrum chart, you can see how it works–the highest intensity is in the visible, but the IR has an awfully long ‘tail.’
https://en.wikipedia.org/wiki/Sunlight#/media/File:Solar_spectrum_en.svg
Anyway, sorry to interrupt. Do carry on.
Lawrence, that change still misses what is going on. Think about it step-by-step: greenhouse gasses being transparent to most of the sun’s incoming energy means some of the UV, all of the visible light, and most of the short-wave IR passes right through them unimpeded (the red area under the left bell curve). That energy is absorbed by earth’s surface, warming it, and the warmer surface then radiates that heat away in the form of long-wave IR light (the right bell curve). Greenhouse gases absorb all but the blue area under the right curve (the atmospheric window). It’s the blocking of this outgoing energy that is the greenhouse effect. By increasing CO2 by 40% we’ve made that blue area just a tiny bit smaller, which has caused the surface to warm by around 1°C (1.8°F) so far.
With that in mind, how about: “The atmosphere contains a few gases that are mostly transparent to incoming sunlight, allowing it to warm earth’s surface, but these gases are opaque to (block?) the heat then radiated by earth’s warm surface. These are called the greenhouse gases.”
It still needs some work, but it better distills the essence of what is going on and which energy is being blocked.
The complete opacity of greenhouse gases to certain wavelengths of long-wave IR is a key concept here. You might find the following anecdote and analogy useful.
Back when I used to post to climate change comment threads at Huffington Post one AGW-denying commenter related that they knew a high school science teacher who illustrated the inconsequential trace nature of CO2 by having his students calculate first the volume of the school swimming pool, and then the corresponding volume of 400 parts per million of the pool’s volume. For illustration let’s assume a 10 meter x 25 m x 1.5 m average depth pool, which works out to 375 cubic meters of water, or 375,000 liters. 400 ppmv would be 150 liters. The teacher then asked his students if they thought adding 400 ppmv of CO2 to the atmosphere could make any difference. The commenter thought this was an oh so clever analogy until I suggested that since CO2’s opacity to IR is the issue he should ask his teacher friend to have his students add 150 liters of India ink, better yet, 150 liters of pure lamp black to the school pool to see if it would make a difference. I’m sure the school phys ed and maintenance staffs would have been none too pleased, but the confused science teacher and his students would have learned something far more insightful than how to do a trivial volume and concentration calculation.
Well blow me down, and here I thought GWA had been shut down and it’s images folded into the wiki commons. Thanks for that, Timothy.
Hank Roberts, #49, your interpretation of my expression “the Suns Heat” is not as I meant. I do not mean it is some special type of Heat, I simply mean “the heat from the sun”. It is unclear if you read my original post (#42), if you had, you would also realise that I have covered the effect of the Sun’s heat when it reaches the ground. I use the comment in the context of a lay audience.
Parts of your reply to me do not seem to make sense. Heat (or thermal radiation) is not just infrared, thermal radiation extends from microwaves all the way to the beginning of the ultraviolet range, this means that all of the visible wavelengths are also part of the Thermal radiation band.
What is the best value for the IR transmittance of the atmosphere?
/1/;40.1/398.2= 0.101
/2/;20/398= 0.05
/3/;40/396=0.101
/4/; 0.126
Sources:
/1/ NASA 2014 ERB-poster-combined-update-3.2014
/2/Stephens 2012 An update on Earth’s energy balance in light of the latest global observations
/3/Trenberth 2009 Earth’s Global Energy Budget
/4/ModTran: Tropical atmosphere, vapor standard, 400 ppm CO2, 1.8 ppm CH4, NOAA Cirrus model
Kevin McKinney @52 wrote that “insolation is ‘majority IR’ by energy” due to the fat tail in the short-wave IR region.
True, hence the necessity to distinguish between short-wave IR and long-wave IR. The sun radiates in the former, earth radiates in the latter. I see the distinction made all the time by climate scientists, but we lay readers seem to gloss over it.
BTW, the solar spectrum Kevin linked to nicely illustrates one reason why H2O accounts for ~80% of the greenhouse effect: it absorbs broadly across both the short-wave and long-wave IR spectrum, while except for one small band, CO2 absorbs only long-wave IR. Thus CO2 only absorbs outgoing energy, while H2O absorbs both incoming and outgoing. This is why in a CO2-forced warming atmosphere the feedback of rising atmospheric H2O more than doubles the warming of the increase in CO2 itself, a concept greenhouse effect deniers simply can not seem to wrap their heads around.
LM 55,
The breakup normally used by radiation physicists is gamma rays around 0.01 microns, X-rays around 0.1, UV to 0.4, visible light to 0.7, near-infrared to 4, thermal infrared to 1215 or so, then microwave, radio, etc. Climate scientists nearly always use “thermal infrared” to mean only 4 to 100+ microns.
Sorry, I meant “125” above, not “1215.”
Barton–“1250”, maybe, if I’m reading the graph right?
K 60,
No, I meant “one hundred twenty-five.” I have also seen 200, 400, and 1000 used as the upper range of “thermal IR.”
Jim,
Re your 57 where you state “H2O accounts for ~80% of the greenhouse effect”, are you aware that only applies in the tropics? In polar regions water is mainly in the form of ice and the concentration of water vapor is negligible. As temperatures in the Arctic rise the greenhouse effect of water vapor will increase and may runaway. Let’s face it, temperatures in the far north can rise monotonically for up to six months!
Some odd comments on here and there
First, a common mistake is to neglect the IR emission from greenhouse gases. Greenhouse gases, as well as absorbing IR radiation, emit it. It gets a bit complicated because almost none of the greenhouse gas molecules that absorb IR light emit it immediately. Instead the internal excited energy of the molecule is transformed into thermal motion of the molecules nearby through collisions. This takes about a microsecond, a millionth of a second and is roughly a million times more likely than the molecule directly emitting IR light.
In the same way unexcited greenhouse gas molecules can be excited by collisions into a state where they emit. It turns out that the rate at which excited molecules can form and their emission spectrum is determined by the temperature, so by looking at the spectrum we can tell the level at which the Earth radiates to space. At the surface there is an equilibrium population of collisionally of about 5% of theCO2 in first excited vibrational state which can emit.
Second, Eli just put up a post on the near IR (NIR) absorption of water vapor and the effects that this has. A key point is that although the NIR absorption populates high vibrational levels of water vapor, these do not emit, because it would require a much higher temperature (like in a hot furnace) for those states to be thermally excited
http://rabett.blogspot.com/2016/02/water-vapor-water-vapor-everywhere-and.html
Alastair, yes, I am well aware of that. Atmospheric H2O is also very low over continental desert regions, all of which goes to show that H2O is not uniformly dispersed in the atmosphere, either horizontally or vertically. We’re talking about the greenhouse effect across the planet as a whole here, and we’re also including liquid H2O in the form of cloud droplets in that 80% figure.
Thank you for your contribution but I missed the part about how to radiate another 7 to 10 Zeta Joules off the planet every year and further human advancement on this planet. Maybe that’s just not that important to scientists. In my freezer, in 2.4 hours I demonstrated that moving ice off the surface radiated about 400 W m-2 more than leaving it covered. Considering the 15 minute tapping may have added a bit of energy I extrapolated to determine by analysis that removing 1% of the polar-night ice extent would add at least 1% extent to summer ice to radiate most all the average annual energy accumulation over the last 40 years. It would be lovely if we could get this unimpressive bit of analysis published. It would be more impressive if we could get a few to support the proposal. http://climatecolab.org/web/guest/plans/-/plans/contestId/1301414/planId/1327806
Mr. Davis, did you account for absorption and reflection of the newly reflected energy in the atmosphere? Radiation from the surface, whether emitted or reflected, does not all make it through the atmosphere.
http://www.asterism.org/tutorials/tut37%20Radiative%20Cooling.pdf
I used the modified Swinback model.
http://i.imgur.com/UKPqI1d.jpg
Polar night sky
Aaron Davis, D. Eng:
Well, keep in mind that the contributors to RealClimate are mostly climate scientists, who with their peers have elucidated how human advancement has caused all those Zetajoules to accumulate in the Earth system. Other scientists, chiefly those specializing in Ecology or Economics, are elucidating the effects the resulting rapid rise in global temperature will have on human advancement, and their findings to date do imply “costs associated with forced migration of 10s of millions of people, abandonment of trillions of dollars of real estate to sea level rise and the devastation of the Arctic environment to heat pollution”, as you observe in your linked proposal. You can be sure that’s as important to scientists as it is to anyone.
To RealClimate scientists and those of us who listen to scientists, it’s more than sufficiently clear that merely maintaining the present state of human advancement (to say nothing of furthering it) requires rapid reduction of the rate of Zetajoule accumulation to effectively zero. Specialists in other scientific disciplines have already done most of the basic research needed for human advancement to continue without accumulating more Zetajoules. Whether that can be done rapidly enough to avoid costs even greater than those you mentioned is up to engineers and policy makers.
Having said all that, your proposal to radiate more accumulated Zetajoules by using icebreakers to expose more open water in the Arctic Ocean during polar winter doesn’t actually seem crazy to me. It’s not self-evident however that your freezer experiments can be scaled up to 1.5 million km^2 quickly and cheaply enough to break even, while competing for resources with the costs and benefits of rapidly halting the accumulation of additional heat, and assuming all costs of both courses of action are internalized.
Lastly: what indefinite human advancement means in a finite Universe is for semanticians, both professional and lay, to ponder.
Aaron Davis, unfortunately your proposal does nothing to address ocean acidification. Excess atmospheric CO2 does more than warm the planet.
The blog entry and the conceptual model are fine, but the paper itself has this inconsequential stray passage, possibly inspired by a critical reviewer: “One objection to the interpretations presented here may be that it is difficult to see how OLR measurements are sufficiently accurate to detect the expected 1W/m2 change in OLR due to CO2…”
This reflects (no pun intended) a separate all-too-common misconception.
Consider the long-time or equilibrium response to greenhouse gas forcing. With no feedbacks, or with feedbacks operating only on longwave radiation, the outgoing longwave radiation must eventually equal its pre-perturbation value. But with positive shortwave feedbacks, such as snow- and ice-albedo feedbacks, net absorbed solar radiation has increased, so outgoing longwave radiation must increase beyond its pre-perturbation value to attain a new equilibrium.
In our time-dependent world, there is a competition between the immediate but temporary decrease in OLR caused by GHG forcing and the slower but permanent increase in OLR caused by shortwave feedbacks. It happens that, with greenhouse gases increasing at their present rate, the overall OLR change is much less than 1 W/m2 (and not necessarily negative) and the top-of-atmosphere energy imbalance is dominated by the feedback-driven increase in net incoming solar radiation.
I say “all-too-common” because I myself clung to this misconception until I read the excellent analysis and explanation of this issue by Donohoe et al. (PNAS, 2014).
Just to follow up on John Nielsen-Gammon’s comment above:
Certainly, OLR can increase with CO2 as the system moves to equilibrium (see also Isaac Held’s post). Though, I found the framing in the PNAS paper to be a bit odd…if you assume shortwave feedbacks are positive, instantly quadruple CO2, and then let the system run for a million years, your equilibrium ASR anomaly is positive (relative to the unperturbed case) and the OLR anomaly is also positive, meaning the system is shedding heat to space more efficiently (despite increased opacity).
If you take a time integral and count the number of accumulated (or depleted) joules, then some might interpret this as the longwave contribution as actually cooling the planet. I find this a rather useless way of conceptualizing the physics of the problem, which is really a tug of war between opacity (say, a reduction of 8 W/m2 at fixed temperature after 4xCO2) vs. all the processes that are conspiring in the column to emit +8 W/m2 (or more). Even without SW feedbacks, the eventual OLR anomaly would be zero after the temperature has increased, and you’d expect to observe something between 0 and 8 W/m2 radiative imbalance from space, depending on when you do the observing relative to the injection of CO2. It is instead helpful to plot the OLR vs. surface temperature while being aware of how ASR depends on surface temperature, such as in these plots.
Also, because of SW feedbacks/forcings, the height of the 255 K isotherm is not really a useful indicator of atmospheric opacity. Any isotherm should move upwards in an atmosphere that is warming. The appropriate “emission temperature” when averaged over all wavelengths should be time-varying, and the link to a surface temperature can be complicated by changing lapse rates.
A follow up to Eli’s comment: At the surface there is an equilibrium population of collisionally of about 5% of the CO2 in first excited vibrational state which can emit.
As I recall the equilibrium situation is that ~5% of the CO2 molecules will be collisionally excited above the first vibrational level. However this energy will be shared among the translational, rotational and vibrational modes so only a small fraction of those molecules will be in the first vibrational excited state. The collision would have to occur at the correct orientation for this to happen.
Benestad & al
The most interesting details here for me were your mentioning of changes in the hydrological cycle as maybe the more important (to humans, to me at least..) consequence of eventual global warming.
The stronger rain all year around is really what I am practically most disturbed by and worried about, allthough it makes us exel in hydroelectric power. But the potatoes allmost drowned last year, and the houses and garden furnitures and fences rot quite much more.
The Dept. of environment has also told it. It will become wetter warmer and wilder they say. So this should rather be taken into conscideration by a lot of people.
Then a little remark by Gavin Schmidt ” ….You see changes from dry adiabatic to wet adiabatic lapse- rate is a negative feedback to increased GHG”
That principle and argument can be understood by 3 principles
§1, The dew point curve of water
§2, Aristoteles theorem: “What goes up must come down again” ( stated before the invention of proper space rockets)
§3, The permanence of matter.
People often forget it when they see warmth leading to draught. Forgetting Aristoteles and the permanence of matter. Whatever vapors off will quite necessarily have to fall down again somewhere.
Thus I hope for the future that water and moisture and the hydrological cycle will be more of your alls concern , because it does entail a lot of further tings for the atmosphere and for the climate and for practical life.
For instance that the climate sensitivity Delta T pr doubling of GHG, given that it can be determined precisely at all, will hardly have any constant value over a wider range of temperatures. Water and the hydrological cycle seems to disturb that.
I guess I should have posted this here rather than in the open thread:
http://bartonlevenson.com/GreenhouseEffect.html
Re Carbomontanus @75:
“The most interesting details here for me were your mentioning of changes in the hydrological cycle . . .”
And (not) nice to see your observations of drowning potatoes and rotting furniture. An actual experience of living in a hothouse.
For as long as I remember, my summers have had many days above 40 degrees, but as it is a dry heat, we manage OK with that. Recent summers, specially this one, when it gets anywhere near 37, it is unbearable, running with sweat and harder to breathe. This takes 4 hours out of my day, so I hide in a hole in the ground, have a siesta and continue my work in the evening. Slightly better, but often still 29 degrees at 11pm.
Is this what some of us can expect to get worse because of the Greenhouse effect ?
“But is it possible to provide a simple description that is physically meaningful and more sophisticated than the ‘blanket around earth’ concept? I wanted a description that could be grasped by physicists. Without the clutter of too much details – just the essentials. A ‘back-of-the-envelope’ type derivation of the greenhouse effect.”
A worthwhile goal and not-so-easy to achieve as we see. A bigger and more important challenge is to come up with an explanation, not for the physicist but for the layman. The “blanket around the Earth” may be the zeroth order attempt but it is very unsatisfactory. Ideas that involve processes are not easy as biologists have found in trying to explain evolution by natural selection to the public. Feedbacks? Lapse rates? Forget it. I think this is an important challenge for Gavin, Rasmus et al. If you can clearly explain why the interior of the car can be so much hotter than the outside air temperature, I think you’ve got it licked and can forego details about convection, varying optical thickness of the atmosphere, etc. Or rather leave those to next level accounts once someone has firmly grasped the car greenhouse effect.
PHIL #74: Since at local thermodynamic equilibrium the same temperature characterizes all of the modes of motion and ~5% in v=1 is what you calculate from the vibrational partition function at T=300K, there will indeed be ~5% in the first vibrationally excited state. Of course, you are perfectly right that the partition function for the rotational and translational modes of motion are also characterized by the same temperature.
Equipartition breaks down above the mesopause, where there are not enough collisions to thermalize any energy deposited into any mode of motion. Of course, there are not many molecules there either.
#28–Alan, maybe I’m missing something here, but IMO the car case is:
1) easy, and
2) not helpful with the atmospheric case.
The main reason for ‘automotive warming’ is the complete elimination of convective cooling–which also renders that case decisively different from the atmospheric case. Comparing the two, again IMO, is only helpful as a contrasting case, not an analog.
The links in the right sidebar — particularly the oddly abbreviated first link under the Science heading — is a good place to start. Much effort has gone into clear explanation, and beginning there rather than reinventing them may save some time and effort.
http://www.aip.org/history/climate/
AD 78,
1. Sunlight makes it through the atmosphere largely unimpaired and heats the ground.
2. The ground radiates infrared light back up.
3. Greenhouse gases in the air absorb infrared light.
4. The greenhouse gases warm up, and radiate.
5. Some of the radiation from the greenhouse gases goes back to the ground.
Thus the ground has both sunshine and “airshine” (atmospheric back-radiation) heating it, and is warmer than it would be without the greenhouse gases.
The atmospheric back-radiation can be measured with a device called a pyrgeometer, in use since at least 1954. In 2004 and 2006, spectral definition studies of the back-radiation showed that the amount coming from greenhouse gases specifically had increased since 1970. This was the smoking gun that it was increased greenhouse gases that had been warming the Earth.
#82–“The atmospheric back-radiation can be measured with a device called a pyrgeometer, in use since at least 1954.”
Yes, and radiational studies, both of insolation and of various inter-atmospheric flows, go back a long, long way farther, as I wrote about in several places:
http://hubpages.com/education/Global-Warming-Science-In-The-Age-Of-Washington-And-Jefferson-William-Charles-Wells
http://hubpages.com/education/The-Science-of-Global-Warming-in-the-age-of-Napoleon-III
http://hubpages.com/education/Fire-From-Heaven-Climate-Science-And-The-Element-Of-Life-Part-One-Fire-By-Day
http://hubpages.com/education/Fire-From-Heaven-Climate-Science-And-The-Element-Of-Life-Part-Two-The-Cloud-By-Night
Kevin-san,
In the essay on Wells, I think you probably mean “radiative cooling” where you have “radioactive cooling.”
#82–Ta, Barton. Self-editing is hell…
Kevin-san,
I know what you mean. Thank God, for my fiction, at least, I have a writer’s workshop. I can go over my own stuff ten times and always miss the glaring error, misspelling, or missing word.