I’ve put together an easy-to-play-with online model of methane in the atmosphere. I’m going to use it for teaching along with the rest of the Understanding the Forecast webmodels, but it was designed to be relevant to the issue of abrupt new methane burps as we’ve been ruminating about lately on Realclimate.
The model runs in three stages: a pre-anthropogenic steady state which ends in the model year
-50, addition of a new chronic source for 50 years (from human activity),then a spike beginning at model year 0 (supposed to be today) and running for 100 years into the model future. Here are results from the “worst case scenario” in the last post (whether you believe it is the true worst case or not): 200 Gton C over 100 years.
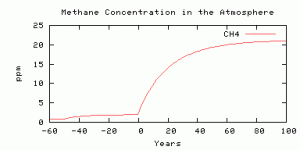
Looks like we got the factor of 10 methane increase about right.
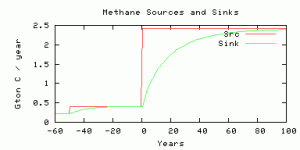
Source and sink of methane in the model.
The lifetime of methane in the atmosphere, used to calculate the methane sink in any time step, is parameterized as a function of concentration following Schmidt and Shindell (2003).
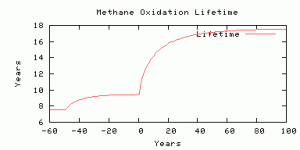
The atmospheric lifetime of methane, used to calculate the sink flux.
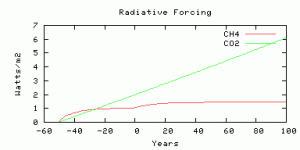
The radiative forcing is parameterized from output from the NCAR model, scaled by an efficacy factor of 1.4 from Hansen et al, (2005). The radiative forcing is compared with Business-as-usual CO2 radiative forcing with the model year 0 corresponding roughly to year 2010, and with CO2 rising at 0.65% per year. The methane radiative forcing before year 0 is not time-realistic because the real human sources did not switch on instantaneously 50 years ago, but you can compare the future evolution of radiative forcing from CO2 and methane, from year 0 onwards.
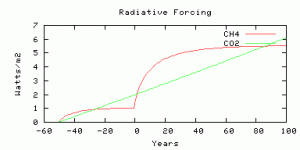
The radiative forcings of CO2 and methane compared. The scenario is more-or-less comparable to 750 ppm CO2, as we thought.
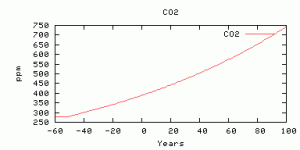
The CO2 concentration used to generate the last figure.
Timing is everything
Four simulations with the same amount of carbon released as methane in the “spike”, on different time scales for the release.
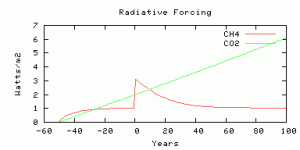
10 Gton C release in 1 year — the spike.
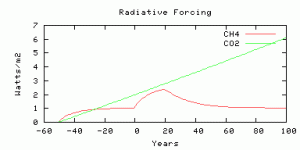
Same spike but not as sharp: 10 Gton over 20 years.
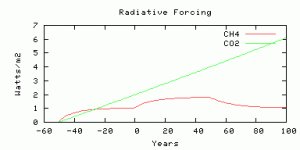
Same 10 Gton but spread over 50 years.
Enjoy. Go and get your swamp gas on, and give the poor model planet your worst. Bwahahahahaha!
References
- G.A. Schmidt, and D.T. Shindell, "Atmospheric composition, radiative forcing, and climate change as a consequence of a massive methane release from gas hydrates", Paleoceanography, vol. 18, 2003. http://dx.doi.org/10.1029/2002PA000757
- J. Hansen, M. Sato, R. Ruedy, L. Nazarenko, A. Lacis, G.A. Schmidt, G. Russell, I. Aleinov, M. Bauer, S. Bauer, N. Bell, B. Cairns, V. Canuto, M. Chandler, Y. Cheng, A. Del Genio, G. Faluvegi, E. Fleming, A. Friend, T. Hall, C. Jackman, M. Kelley, N. Kiang, D. Koch, J. Lean, J. Lerner, K. Lo, S. Menon, R. Miller, P. Minnis, T. Novakov, V. Oinas, J. Perlwitz, J. Perlwitz, D. Rind, A. Romanou, D. Shindell, P. Stone, S. Sun, N. Tausnev, D. Thresher, B. Wielicki, T. Wong, M. Yao, and S. Zhang, "Efficacy of climate forcings", Journal of Geophysical Research: Atmospheres, vol. 110, 2005. http://dx.doi.org/10.1029/2005JD005776
I note a very readable article in the February GSA Today by Kidder and Worsley outlining a progression from “icehouse” through “greenhouse” to “hothouse” via a HEATT (haline euxinic acidic thermal transgression) episode. In this progression, the “thermal mode” haline circulation via polar sinking of cold brines gives way to a “haline mode”, of sinking warm brine driven by evaporation, together with increasing anoxia and euxinia. The authors conclude that we are probably near a “cool greenhouse” where most northern ice melts and half of Antarctic ice sheet,but that it would be difficult to create a “hot greenhouse” or a “hothouse” even if we burn all the fossil fuels, and that the “icehouse” will not return for millennia.
sidd
Hi All-
David says a worst case release from the hydrates is about 200 Gton of carbon from the methane hydrates. Here’s a paper on two events during the Jurassic which talks about 3000-5000 Gton releases of methane, in order to fit their modeling of the carbon isotope excursions during these periods. That’s 15 to 25 times as much methane- doing the math.
ON THE NATURE OF METHANE GAS-HYDRATE DISSOCIATION DURING THE TOARCIAN AND APTIAN OCEANIC ANOXIC EVENTS
Leland (~#302), you continue to make unscientific extrapolations. The Beerling et.al. article you cite is 10 years old and only states that their findings are consistent with their hypothesis, and another Beerling & Berner paper in the same year claims results provide a preliminary theoretical explanation of their idea. A quick forward search of more recent research that cites these papers demonstrate other mechanisms for catastrophic methane and CO2 release not related to warming. Examples:
2005- http://www.sciencedirect.com/science/article/pii/S0012821X05003730
2007- http://folk.uio.no/hensven/Svensen_etal_EPSL_07.pdf
2010- http://www.sciencedirect.com/science/article/pii/S0016703710005156
What has Beerling been saying more recently about large releases of methane from global warming? What evidence do you have of any kind of consensus on this question? This is a science, not a speculation forum. A current review article that supports your claims would be useful.
Did you actually read the good 2011 review in Nature linked by Hank Roberts above (#184). There are a lot of good references there:
http://www.nature.com/scitable/knowledge/library/methane-hydrates-and-contemporary-climate-change-24314790
Some further quotes from this review:
“4. Deepwater gas hydrates (~95.5%).
These gas hydrates, which constitute most of the global inventory, generally have low susceptibility to warming climate over time scales shorter than a millennium.”
And:
“Conclusions
Catastrophic, widespread dissociation of methane gas hydrates will not be triggered by continued climate warming at contemporary rates (0.2ºC per decade; IPCC 2007) over timescales of a few hundred years.”
But also:
“Proof is still lacking that gas hydrate dissociation currently contributes to seepage from upper continental slopes or to elevated seawater CH4 concentrations on circum-Arctic Ocean shelves. An even greater challenge for the future is determining the contribution of global gas hydrate dissociation to contemporary and future atmospheric CH4 concentrations.”
This is a balanced look at the methane problem and it does not support your alarmism. Please provide some scientific evidence to back your assertions, not opinions. Steve
> provide some scientific evidence to back your assertions
Wait, it sounds like Leland’s fallen into that trap called ‘reverse citation’ — it’s a bad practice.
That’s not what Steve is suggesting, as he makes clear.
But for Leland — seriously — think about whether you’re starting from what you believe and scrounding around for stuff you can claim as evidence.
It doesn’t work out well.
It’s an Internet-era problem, where students will write down whatever it is they believe, then ‘oogle the buzzwords and pick and choose to get “citations” they can use for references.
Before the Internet, it was easier to search for sources first and work from those to reach conclusions — because it’s so much more work to search through vast amounts of literature to pick out a few sources that can be claimed as support for some odd outlier belief.
Now — it’s just as easy to find “citations” for odd beliefs because ‘oogling does the work of searching through all the other reference material.
Don’t start with assertions and then go look up ‘scientific evidence’– because you can find anything on the Internet.
Either go to a good reference librarian in that area of science, someone who knows the literature and how to search it — or start from good review articles and follow citations forward.
That’s what Steve did there, starting from the review article I mentioned.
Leland, looking back a bit, you were on this same subject at
http://thinkprogress.org/romm/2009/08/17/204508/positive-methane-feedbacks-permafrost-tundra-methane-hydrates/
citing a web book “killerinourmidst” dated 2004-2007
The author of that web book cites as his primary source: David Archer.
Here, you’ve been reading newer information from David Archer, and you keep complaining that he’s not doing it right, based on — old information.
Do what Steve points out — follow citing papers forward and see what’s being made of an idea as later work is done on it.
And be very wary of commercial interests that will start hyping anything that they can make money from — like “pressure relief” by drilling for gas.
Hi Hank-
My criticisms of David’s online model have to do with factually correct concerns.
Firstly, he leaves out secondary CO2, produced by methane oxidation. For small releases of methane, this is OK. For large releases of methane, this is actively misleading, and gives a mistaken impression or conceptual frame of the duration of the crisis. Secondary CO2 could also lead to new rounds of deeper hydrate dissociation. The graphs at the top of this page give the misleading impression that substantial releases of methane could be quickly recovered from. This is unlikely, at best.
Secondly, in a companion article to the online model, he suggests that 200 Gtons of methane evolved over one hundred years constitutes a worst case scenario. There are hundreds of scientific papers, recent as well as a decade or more old, which calculate methane releases much greater than that- on the order of 15 to 25 times as great. Most of those releases are believed to be over a longer period of time than one hundred years- but our triggering blast of CO2 is also being produced much more rapidly than past triggering events.
Thirdly, he includes business as usual CO2, methane, and increased methane lifetime, but leaves out secondary atmospheric effects of methane, as modeled by Isaksen, for example. There is no provision for inclusion of stratospheric water vapor, tropospheric ozone, or stratospheric water vapor in David’s model. Any model claiming to be a worst case scenario should include these.
Fourthly, David leaves out oceanic chemistry effects of methane. Modeling from LBL and LANL suggests that large sustained releases of methane would start to exhaust the methane oxidation ability of the Arctic ocean, and lead to methane bubbling directly into the atmosphere. Only by including steadily increasing quantities of methane into the inputs to the model, something that the model does not allow, can this oceanic chemistry effect be included.
Fifthly, David does not include the oceanic chemistry effects of methane on ocean production of nitrous oxide. Recent studies have indicated that oxygen depleted oceans can produce nitrous oxide at rates much higher than those of oxygenated oceans.
In addition, I am not satisfied that David’s model includes much stronger water vapor feedback from all of these sources of forcing.
We don’t even know what’s going to happen. How can anyone, no matter how scientifically astute, predict the outcome of such a snarl of interconnected positive feedback loops, including physical and chemical effects, and come out with a prediction of what the worst case scenario would be, with any sort of scientific credibility?
It’s not really possible, I think.
David’s model needs to include secondary CO2. It needs to be adjusted to allow more flexible inputs of methane, to account for oceanic chemistry effects and exhaustion of Arctic ocean oxidation. It needs to deal with the water vapor feedback in some more straightforward and transparent manner, I think. It needs to allow for the inclusion of nitrous oxide, stratospheric water vapor, tropospheric ozone, and the stronger water vapor feedback from each of these to be a credible worst case model.
My main problem with the model is that this is presented as a worst case model, when it is obviously incomplete, and for large releases of methane is actively misleading.
Correction:
Make that …stratosperic water vapor, tropospheric ozone, or stratospheric hydroxyl radical…
Another problem with David’s model- and with any worst case model- is the logarithmic nature of greenhouse gas forcing.
This effect guarantees that the side effect greenhouse gases, such as ozone, nitrous oxide, tropospheric ozone, stratospheric water vapor, and stratospheric hydroxyl radical will be by far the most potent.
This means that unanticipated side effects can be very, very important, and this undermines the whole idea that it is scientifically possible to come up with a worst case scenario.
[Response: Sorry, but this just isn’t true. OH isn’t a greenhouse gas anywhere in the atmosphere, and in the stratosphere it is not even the limiting factor on CH4 oxidation. Your whole argument appears to be that you anticipate that unanticipated side effects will certainly be “very, very important”, but the effects you mention are neither unanticipated nor unquantifiable. And when they are quantified, they are small. Maybe something unknown will have a big effect – that can’t be ruled out – but your certainty that such a mechanism is not only present and will be dominant has no evidentiary basis. This is why people are not taking your points on board, it has nothing to do with anything else. – gavin]
Hi Gavin-
About OH- well, you’re right. I misread this paragraph from Isaksen:
So while there are massive atmospheric chemistry changes associated with methane atmospheric chemistry, increased forcing by OH is not one of them. I was wrong, and I apologize about that one.
About the future being sufficiently quantifiable to bet the future of the world on business as usual, no, I don’t believe that is the case. The logarithmic nature of greenhouse gas forcing makes low concentration greenhouse gases inhabiting unoccupied absorption bands the most potent. Methane, although you have described it as a “sucky” greenhouse gas (I think that was you, if I remember right) provides about 25 percent as much radiative forcing as CO2 right now, even though its concentration is 150 times less, as you know, due to this logarithmic effect.
Therefore unanticipated side effects could be very important. You’re telling me you have anticipated and quantified every possible interaction, with sufficient confidence to bet the future of the biosphere on business as usual, or even with sufficient confidence to publish a scientifically valid worst case scenario?
Pull on the other leg for a while, this one’s coming off at the hip. :)
Leland @ 309:
Could you please connect your phrases — “About the future being sufficiently quantifiable to bet the future of the world on business as usual . . .” and “. . .with sufficient confidence to bet the future of the biosphere on business as usual . . .”– to anything Gavin or anyone else here has said or implied? Thanks.
What David Archer wrote:
“… The radiative forcing is compared with Business-as-usual CO2 radiative forcing …”
What Leland Palmer seems to believe that meant:
“… bet the future of the world on business as usual ”
Mr. Palmer — “business as usual” is _described_ not _recommended_ above. You focus on the unlikely — and ignore and so minimize the likely.
Give us a break to talk about the science for a while, eh?
Leland, I’m beginning to think that the solution to your problem may have less to do with atmospheric chemistry and more to do with phar_ma_cology.
Hi Ray-
Oh, thank you. :)
Here’s another interesting oceanic chemistry process, not taken into account by David’s “worst case scenario”- methane driven denitrification:
Funny, I didn’t see increased production of nitrous oxide anywhere in David’s “worst case scenario”. Yet, large releases of methane have been associated with anoxic oceans in several past apparent methane catastrophes. The modeling by LBL and LANL also shows this, of course.
David’s model is a very simple one. Can such a simple model, which leaves out chemical and biological effects of methane release, possibly claim to be a “worst case scenario”?
[Response: a) this has nothing to do with methane. b) why you appear to think that repeating your point for the 20th time will make a difference when the previous 19 times didn’t remains a mystery. – gavin]
Link to the above:
Oceanographer: Nitrous Oxide Emitting Aquatic ‘Dead Zones’ Contributing To Climate Change
Actually, gavin, it has everything to do with releases of methane.
Oxidation of methane to CO2 uses up available oxygen in seawater. Ocean suboxia and hypoxia are known effects of methane release. Ocean anoxia and acidification have been associated with past apparent methane catastrophes. Not to mention that global warming itself could disrupt global thermohaline water circulation, leading to widespread anoxia and hypoxia
From Lawrence Berkeley Labs, and their study of methane release:
As Climate Changes, Methane Trapped Under Arctic Ocean Could Bubble to the Surface
[Response: Sorry, but no. The oxygen minima zones have no relation to areas of methane release, and the quote you pulled in your last comment didn’t mention methane once. The quote now demonstrates that there isn’t much capacity to in the ocean to soak up much methane (since the volume affected by a plume is small and easily saturated), so the idea that sea floor plumes would lead to widespread anoxia (away from where the actual plumes are) is not supported. You are simply stringing together disparate bits of science that use similar words but in different contexts in order to spin a fantasy of imminent catastrophe. It is a great shame that the fact that you appear to be getting the impression that I don’t think any of this stuff is interesting – nothing is further from the truth – but making progress means paying attention to the details and actually quantifying the size of various effects – and explaining why none of this happened in the Early Holocene, the Last Interglacial, the Pliocene, or indeed any time in the last 50 million years. – gavin]
Leland, this is getting into Gish territory now. Please, give it a rest. The more you say, the less convincing (or even interesting) you become.
Making unsupported alarming statements, refusing to cite research to support ones claims when asked, citing only cherry picked research, ignoring substantive criticism, writing long and repetitive posts, and the willingness to criticize experts when one has no expertise themselves are all trolling behaviors. I believe that a series of posts that continue this behavior for too long should go straight to the Bore Hole.
On the other hand, I also believe that if I were in charge the world would all be like “The Big Rock Candy Mountain.” Steve
Tangential tidbit, from our local earthquake response info — consistent with earlier stories about the overall leakiness of the natural gas distribution systems around the world:
“Many of the older homes in the area already have tiny gas leaks now that might not be detectable by your nose. You can purchase a sniffing device in advance of a disaster and sniff the entire house for leaks. That way, you’ll know if the house already has a problem ….”
Hi gavin-
Let’s try this again. Perhaps a shorter reply will get through moderation.
One problem with David’s “worst case scenario” article and the accompanying online model is that neither of them contain any chemistry, of any sort, either atmospheric or oceanic.
Does he really expect that we can dump billions, tens of billions, or hundreds of billions of tons of methane into the oceans and atmosphere, and have no chemical effects?
Massive dissociation of gas hydrate during a Jurassic oceanic anoxic event
So, once again, these guys are talking about trillions of tons of isotopically light carbon, from methane hydrates- 1.5 to 2.7 trillion tons, they say. More methane than that is required, though, if synchronous burial of C12 enriched organic carbon is taken into account.
And, they are associating this with an oceanic anoxic event, and with methane oxidation.
Would suboxic conditions result in increased production of nitrous oxide, perhaps vastly increased production?
[Response: No-one is arguing that a massive methane release would have no effects on anything. The issue is whether anything that is conceivable now (not in the Jurassic, not in the Eocene, not at the PT event), not over 60,000 to 100,000 years, but over decades, that would radically alter any near term trajectory. If your point is simply that a 10,000-fold increase in methane emissions is a worse case scenario than an 100-fold increase in methane emissions, I’m not sure who you are arguing with. If you think that this increase in methane emissions is remotely credible, then we are just going to have to agree to disagree. – gavin]
Hi gavin-
Well, if such massive methane releases have happened before, not once but maybe a dozen times, then as a worst case scenario, they are credible, don’t you think?
[Response: Snowball Earth happened before, so by this argument it is credible that it could happen tomorrow. Sorry, but I don’t buy it. The Arctic has been warmer than today probably in the Early Holocene, certainly in the Eemian, at Stage 11, the Pliocene etc. But no huge methane spike has occurred in over 50 million years. Thus if you want to make the case that it is credibly imminent, you have to explain why it didn’t happen then. – gavin]
Group think has set in on this site, in my opinion. You guys seem to have come to some sort of consensus that it is possible to construct a worst case scenario before we even know what will happen qualitatively. I don’t think it is possible to quantitate a situation when we don’t even know what will happen qualitatively.
There are several factors which might make the methane releases faster, this time. Also, even if the crisis does not develop faster than it has during past methane catastrophes, it might be essentially unstoppable, even with advanced technology and a true war on climate change. So even if it doesn’t happen in a couple of decades or a century, it may certainly be that our window of opportunity to stop a methane catastrophe is rapidly closing.
Firstly, the sun is hotter than it was during the End Permian, by a couple of percent, according to the standard model of stellar evolution. According to Hansen, that is equivalent in forcing to maybe 1000 ppm of CO2.
Secondly, our triggering blast of CO2 is unnaturally swift, and unnaturally systematic. Past triggering events, by the clathrate gun hypothesis, have very likely been slower and very likely much more random.
The positive feedback nature of some of these feedback loops are also very worrisome, of course.
Our geological situation could also be less stable than in past methane catastrophes. It may be that having a region like the East Siberian Arctic Shelf, with its shallow subsea permafrost and shallow hydrates, has never happened before. Due to the positive feedback nature of methane release, and the ancillary atmospheric and oceanic chemistry effects that David’s “worst case” model neglects, this alone might accelerate our coming methane catastrophe beyond what has occurred before.
David could do us a real service by revising his model, I think. Unfortunately, if he does that, he will likely have to revise his worst case scenario upward.
[edit – just stop with personal comments]
Leland,
For the umpteenth time: The model may not explicitly do atmospheric chemistry, but it is implicit in the parameterization of the ‘efficacy’ of methane (the 1.4 factor). This comes from Hansen et al., p. 38, and accounts for the direct methane forcing as well as indirect effects certainly including stratospheric water vapor and tropospheric ozone (but not the secondary CO2).
The methane input is not generated by the model, but by user input, and so is whatever we choose to make it, and includes whatever multipliers we choose to imagine. The model welcomes you to try a different number than David’s 200 GtC. You can release, say, 5000 GtC if it makes you feel better. Kids, don’t try this on your home planet.
The water vapor feedback is dealt with in an entirely transparent manner. It’s not dealt with. It doesn’t have to be. We’re comparing forcings, not calculating temperature change.
#320–
“Group think has set in on this site, in my opinion.”
Why? Because you fail to convince?
Hi gavin-
Sure, no problem.
Rate of change.
Non-random forcing.
Accumulation of methane hydrates during recent ice ages.
Approaches to past warm periods have been gradual, and not systematic. There was a certain amount of randomness mixed in. Methane released quickly has nonlinear atmospheric and oceanic chemistry effects which overwhelm oceanic and atmospheric oxidation mechanisms for methane. This makes methane released quickly much, much worse than methane released gradually.
Past forcing has been mostly orbital based, with some amplification from CO2. What we are seeing now is not global warming as usual- it is unnaturally swift and systematic CO2 based forcing, far different from past gradual orbital variation driven forcing.
We’re told, by David among others, that current methane hydrate stocks are lower than can plausibly be the case for past apparent methane catastrophes. Aren’t we coming out of an ice age, with cold ocean water temperatures? Does this make any sense, at all, that current methane hydrate mass would be so low? Have the fundamental bacterial and geochemical cycles that higher lifeforms inhabit changed in some way, that would explain this?
Leland, here’s a clue. When Gavin asks you to explain why something didn’t happen, he’s not asking you to throw a bunch of stuff at the wall and see what sticks. Do some math.
Hi Ray-
Thank you for your advice. :) Here’s some advice for you- do a little chemistry, specifically atmospheric and oceanic methane release chemistry. Then apply some math to that. :)
Here’s some other math for you, Ray:
From Down the Rabbit Hole:
If there are 5000-20000 Gt of hydrates now and in the past, then past releases of methane of several trillion tons are easy to explain. The isotope evidence of past apparent methane catastrophes and oceanic anoxic events then has a natural and logical explanation.
There seems to be no convincing reason that methane hydrate total mass should be lower than in the past. We’re coming out of several ice ages, Ray, with low water temperatures.
So, if the methane hydrate dissociation explanation for past methane catastrophes is correct, current methane hydrate total mass cannot plausibly be as low as Archer says it is, in my opinion.
[edit – there is no point whatsoever in you continuing here if you keep making ad hom attacks on the authors, their co-authors, uncles, cousins or postmen. Just stop it or leave.]
Leland, listing numbers is not math. You need to show either that such events have happened with some regularity in the past (good luck) or that the situation now poses unique risks IN THE PRESENT TENSE. Handwaving is not science, and you are doing so much of it you’re on the verge of levitating
Leland, if you look back through the years, I have been one of the most forceful–some would say strident–advocates of forceful action. I do so, however, on the basis of known, credible risks. Those alone are sufficiently daunting that anyone capable of intelligent risk assessment will have already realized the necessity of action. Unfortunately, to paraphrase Adlai Stevenson, we need a majority.
The majority of people have no understanding of science or of risk calculus. To convince them, we have to rely on our credibility. That means being right more often than wrong, but it also means we have to be damned sure we aren’t crying wolf. If we do, we play right into the hands of denialists who seek to portray us as “alarmists”. The established risks are alarming enough for anyone who cares about the futures of their progeny.
The risk you have adopted as your own pet cause is a possible risk. I is not as of yet a credible risk–and all the “Well, it could…” or “But it might…” you can muster cannot change that. Only hard facts and evidence matter in establishing the credibility of a risk. I do you no favors if I lower the bar for you. You would merely stumble over the next hurdle. Get the evidence, and then we’ll talk.
Hi All-
It’s kind of instructive to enter 12000 tons of methane released over 10,000 years into David’s model.
Of course, secondary CO2 in David’s model does not change, as we’ve discussed, and in this case this is extremely misleading, because truly massive amounts of secondary CO2 would be produced. But, the model makes this look kind of survivable.
But, suppose our hotter sun, more rapid triggering event, and the unnatural nonrandom nature of our current forcing makes all of this happen in a thousand years. Certainly, most experts doubt that David’s estimate of total hydrate stocks is correct. It seems awfully low, and there seems to be no compelling reason that current total hydrate stocks would be so low.
So, nobody knows how much hydrate is down there, really. Let’s say that it is close to 20 trillion tons, in line with traditional estimates from a decade ago, as a worst case scenario.
Let’s say it takes 1000 years to release it, as a worst case scenario.
Methane concentration in the atmosphere would increase to 180 ppm, over a hundred years, as a worst case scenario.
Forcing, just from methane would increase to about 12 W/m2 over a hundred years, not of course including ozone and stratospheric water vapor, and secondary CO2.
Now, that’s what I call a worst case scenario, with a couple of caveats, of course.
This would be “somewhere over the rainbow” so to speak, in totally uncharted territory. Nobody knows what strange and wonderful atmospheric and oceanic chemistry effects would occur. Ocean anoxia seems likely. Ozone at high concentrations down to ground level in the tropics seems likely, extrapolating from Isaksen. Destruction of stratospheric ozone by stratospheric water vapor seems likely.
I keep wondering about Hansen’s remark “at some point the oceans would start to boil…”. At the current time, I’m not sure that doing all the math in the world will tell us if this will happen, or not, if this more realistic worst case scenario- complete with all of the unknown atmospheric chemistry, oceanic chemistry, and biological effects occurs.
Perhaps a shorter post will make it through moderation:
This paper talks about 12 trillion tons of light carbon from the hydrates, entering the carbon cycle during the triassic mass extinction.
Twelve trillion tons is a big number- 60 times bigger than David’s “worst case scenario”, although admittedly over a much longer time span.
Correction: make that 12000 Gigatons, or 12 trillion tons of carbon, not 12,000 tons.
Sorry.
Whoops, forgot the link-
Atmospheric Carbon Injection Linked to End-Triassic Mass Extinction
Leland, since your numbers are all apparently rectally extracted, why stop at 12000 Gtons?
Hi Ray-
Oh, that number came from a peer reviewed article in Science, the official journal of the American Association for the Advancement of Science, (the AAAS), Micha Ruhl, et al,Science 333, 430 (2011).
They got it by an analysis of carbon isotope ratios. That’s the amount that they calculated would be necessary to cause the negative carbon isotope excursion they observed, occurring at the same time as the End Triassic mass extinction event.
If 12 trillion tons of hydrate methane came out of the hydrates back at the end of the Triassic, and fundamental geochemical and bacteriological conditions have not changed much, it’s kind of hard to see how total methane hydrate stocks worldwide could be 700-1200 Gtons now, at least for me.
I suppose it’s possible, but absent some compelling reason why methane hydrate stocks are so low, right now, Occam’s Razor says that worldwide hydrate stocks should be in the 20,000 Gton range, or so.