In January, we presented Lesson 1 in model-data comparison: if you are comparing noisy data to a model trend, make sure you have enough data for them to show a statistically significant trend. This was in response to a graph by Roger Pielke Jr. presented in the New York Times Tierney Lab Blog that compared observations to IPCC projections over an 8-year period. We showed that this period is too short for a meaningful trend comparison.
This week, the story has taken a curious new twist. In a letter published in Nature Geoscience, Pielke presents such a comparison for a longer period, 1990-2007 (see Figure). Lesson 1 learned – 17 years is sufficient. In fact, the very first figure of last year’s IPCC report presents almost the same comparison (see second Figure).
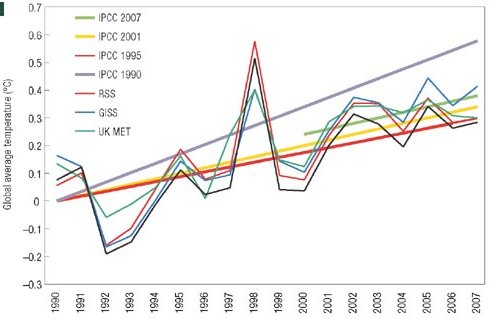
Pielke’s comparison of temperature scenarios of the four IPCC reports with data
There is a crucial difference, though, and this brings us to Lesson 2. The IPCC has always published ranges of future scenarios, rather than a single one, to cover uncertainties both in future climate forcing and in climate response. This is reflected in the IPCC graph below, and likewise in the earlier comparison by Rahmstorf et al. 2007 in Science.

IPCC Figure 1.1 – comparison of temperature scenarios of three IPCC reports with data
Any meaningful validation of a model with data must account for this stated uncertainty. If a theoretical model predicts that the acceleration of gravity in a given location should be 9.84 +- 0.05 m/s2, then the observed value of g = 9.81 m/s2 would support this model. However, a model predicting g = 9.84+-0.01 would be falsified by the observation. The difference is all in the stated uncertainty. A model predicting g = 9.84, without any stated uncertainty, could neither be supported nor falsified by the observation, and the comparison would not be meaningful.
Pielke compares single scenarios of IPCC, without mentioning the uncertainty range. He describes the scenarios he selected as IPCC’s “best estimate for the realised emissions scenario”. However, even given a particular emission scenario, IPCC has always allowed for a wide uncertainty range. Likewise for sea level (not shown here), Pielke just shows a single line for each scenario, as if there wasn’t a large uncertainty in sea level projections. Over the short time scales considered, the model uncertainty is larger than the uncertainty coming from the choice of emission scenario; for sea level it completely dominates the uncertainty (see e.g. the graphs in our Science paper). A comparison just with the “best estimate” without uncertainty range is not useful for “forecast verification”, the stated goal of Pielke’s letter. This is Lesson 2.
In addition, it is unclear what Pielke means by “realised emissions scenario” for the first IPCC report, which included only greenhouse gases and not aerosols in the forcing. Is such a “greenhouse gas only” scenario one that has been “realised” in the real world, and thus can be compared to data? A scenario only illustrates the climatic effect of the specified forcing – this is why it is called a scenario, not a forecast. To be sure, the first IPCC report did talk about “prediction” – in many respects the first report was not nearly as sophisticated as the more recent ones, including in its terminology. But this is no excuse for Pielke, almost twenty years down the track, to talk about “forecast” and “prediction” when he is referring to scenarios. A scenario tells us something like: “emitting this much CO2 would cause that much warming by 2050”. If in the 2040s the Earth gets hit by a meteorite shower and dramatically cools, or if humanity has installed mirrors in space to prevent the warming, then the above scenario was not wrong (the calculations may have been perfectly accurate). It has merely become obsolete, and it cannot be verified or falsified by observed data, because the observed data have become dominated by other effects not included in the scenario. In the same way, a “greenhouse gas only” scenario cannot be verified by observed data, because the real climate system has evolved under both greenhouse gas and aerosol forcing.
Pielke concludes: “Once published, projections should not be forgotten but should be rigorously compared with evolving observations.” We fully agree with that, and IPCC last year presented a more convincing (though not perfect) comparison than Pielke.
To sum up the three main points of this post:
1. IPCC already showed a very similar comparison as Pielke does, but including uncertainty ranges.
2. If a model-data comparison is done, it has to account for the uncertainty ranges – both in the data (that was Lesson 1 re noisy data) and in the model (that’s Lesson 2).
3. One should not mix up a scenario with a forecast – I cannot easily compare a scenario for the effects of greenhouse gases alone with observed data, because I cannot easily isolate the effect of the greenhouse gases in these data, given that other forcings are also at play in the real world.
re Gavin’s comment on tom w.’s #298: I’ve been following this little set-to but don’t wish to enter the fray. I do have a 101 question that you can help me with. Are you saying that Tom’s “icecap” graph was cherry-picked because it starts at the off-the-wall 1998 high temperature point? One other (same graph): I’m curious why the lower trop MSU satellite measurements swing much more violently than HadleyCRUT3v. A simpleton answer will suffice. O.K. — three! Did CO2 concentration really go from 366ppm to 386ppm in ten years?
These are not leading/trick questions…
[Response: 1) Yes – but don’t forget the implicit scaling of the CO2 to temperature (implying about a 12 deg C climate sensitivity). 2) In the tropics, LT varies by 20 to 40% more than the surface changes due to variations in the moist adiabat. This is true on all time scales (monthly, yearly decadally) in the RSS data and the models (but only on short time scales in the UAH data). 3) Yes. Average growth rate is now above 2ppm/year. – gavin]
re gavin’s answer to 301, so the graph I presented http://acute.ath.cx/BULK/global_warming/2008icecap.us-MonthlyCO2vsTemps.jpg is cherry picked. Fascinating, I chose it because it is one of most recent I’ve found and at the source it came from, the data used to create it is also linked. But the fact the data shows the most current decade and it is showing the impact of the last ten years and all preceding decades of the current cusp of doom levels of CO2 doing their year after year heat trapping, it should have no meaning as it is cherry picked.
Such astounding scientific analysis
And for one more funny observation. A cherry picked observation. In general when there is the annual mona loa CO2 peak, there is opposite low in the satellite data.
Clearly CO2 is causing global warming by the law of all data to the contrary is cherry picked and should be ignored.
[edit for tediousness]
Dear Ray Ladbury re 300, Could you give me an example of what you consider pontification. I thought I only posted logic and reason using the laws of physics as I understand them. Did I use them wrong? I do express my opinions on what I interpret data to mean. And when I believe my interpretation is correct, I make that clear. But I am willing to listen to anyone explain how my interpretation is not certain or wrong. The concept of cherry picking to me is alien. And like most who have decades of researching, I find I often using my tacit abilities know when I have been pointed too samo samo hand waving explanations that some may believe are science. I have no doubt that all over the internet one can find CO2 skeptic analysis that is lame and bogus. But I have found all kinds of lame and bogus CO2 gloom and doom analysis at realclimate.org and many others.
hoping one will find the informed who are willing to take time and explain may be quite foolish of me. You see if one really understands clearly, it is quite simple to explain. I know what I see clearly, I explain it as clearly as I can and all the brilliant informed tell me is that I am blind, I am a troll, I am paid agent to make trouble.
I’m a boyscout, OK a seascout. OK I’m a past skipper of a Seascout Ship and a current mate voluteer with that ship. Not By Bread Alone
http://acute.ath.cx/SHIP110/
Does anyone care about finding truth. Does anyone care that if policy drives food to become fuel, Food will become more expensive and in places where food is always short, the carbon footprint of hundreds of thousands if not millions will be eliminated by malnutrition and the pestilence that goes with such malnutrition.
tom watson — let me explain a bit about radiation physics, and then please answer a question for me.
The flux absorbed by the climate system is
F = (S / 4) (1 – A)
where S is the solar constant, the amount of sunlight falling through a unit area at Earth’s mean distance from the sun. S has been measured by satellites at about 1,366 watts per square meter.
A is the bolometric Bond albedo, the amount of sunlight the Earth reflects away from itself in all directions. A is about 0.306 according to NASA.
The factor of 1/4 is because Earth receives sunlight on its cross-sectional area (π R2), but radiates heat from its total area (4 π R2).
The flux absorbed by the climate system then works out to be about 237 watts per square meter on average. We can find out what “radiative equilibrium temperature” this corresponds to by inverting the Stefan-Boltzmann law for radiative intensity:
F = σ T4
where F is the flux emitted by a blackbody radiator, σ is the Stefan-Boltzmann constant (about 5.6704 x 10-8 in the SI), and T the absolute temperature. Solved for T, this becomes:
T = (F / σ)0.25
So Earth’s radiative equilibrium temperature works out to about 254 K.
Water freezes at 273 K. If the Earth’s surface temperature were 254 K, the Earth would be frozen solid. But the Earth’s surface temperature averages 288 K, 34 K higher than the equilibrium temperature.
If this is not due to the greenhouse effect, where is the higher temperature coming from? Where does the energy come from that keeps Earth at habitable temperatures? Sunlight by itself isn’t enough to do it.
Tom Watson, Your post is disingenuous. You have been directed to several resources that detail the physics. Yet you persist in your odd straw-man theories, showing no evidence that you have even perused any of these references. Examples of your incorrect science:
1)assertion that CO2 does not have sufficient heat capacity to warm Earth (heat capacity is irrelevant to the radiative physics involved)
2)your assertion that heat loss of Earth–an isolated system surrounded by vacuum–is dominated by convectiuon
3)You have taken data out of context–e.g. using the 1998 El Nino as a peak of warming
and on and on.
Worst of all, Tom, there’s no evidence your learning curve has a positive slope. Until you make an effort to learn the real science so that you can talk apples with apples, you are wasting our time and yours.
Dear Barton Paul Levenson 304.
I fully understand and do agree with the equations and your explanations of radiation physics.
And they are a very simple explanation of, what I would call “a” driver of the long term average temperatures of the Earth.
Now in a loose sense you use the term greenhouse effect. Or maybe you imply greenhouse effect is only the effect of radiation physics in the termperature of the Earth.
Other effects are not considered part of the greenhouse effect. In this I don’t fully comprehend the intention of your post.
From this one could the define CO2 greenhouse radiation effect as the effect of CO2 absent all other gasses.
From this one can define CO2 it a 20% effect and H20 is a 80% effect. This is the only explanation I have found at realclimate.org.
And maybe I missed it, but all other links I have been given have not expanded on this explanation.
Now all of the above is true, but I comprehend or consider as meaningless and misleading in any search for what real or actual effect CO2 has
at 380 or even 720 PPM. This 1 to 5 relationship ignores the reality of 10 to 50 fold more H20 molecules and that H20 is a
total and partial absorber of far more wavelengths than CO2, and also H20 is a partial absorber of the far fewer CO2 wavelengths that are
in the black body radiation spectrums of the earth’s temperatures.
I have spent much time looking for and when possible annotating images to emphasize what seems obvious to me.
http://e6.ath.cx/gw/Atmospheric_Transmission-sm.jpg
http://e6.ath.cx/gw/Global_Warming_Not_From_CO2-fig1-n-2.jpg
Are all the equations of the models that compute CO2 driving temperature using the above weightings?
Then I may know why the models predict manifestations that cannot be found.
The atmosphere is a moving, sometimes violently moving fluid, that is also why I also opine the equations that are really about static
homogenized static fluids have little reality in suggesting some rules of actual reality.
A good representation of the simple average of all is this. This is how I consider and what I consider the greenhouse effect.
What part is radiation physics and what part Newtonian physics. Can one separate them.
http://e6.ath.cx/gw/2007.nasa.gov.energyballance.gif
Ray, a quicky out of left field (well…actually right field, I guess…): Is heat capacity really irrelevant to the radiation physics? Doesn’t radiation energy go to the same molecular place that latent heat (energy) goes, and doesn’t that have some effect or relevance?
Rod B, Radiation excites those modes for which it has the appropriate energy–independent of temperature (to first order, anyway). Heat capacity depends on temperature, because it depends on which modes of the molecule are thermally excited. For a very cold gas, the vibrational modes are pretty much irrelevant to the heat capacity. As you raise the temperature, there are more modes into which energy can go, so the heat capacity (amt of energy needed to raise temperature) rises. Remember that temperature is a measure of the average energy per degree of freedom of the gas.
Tom Watson, if you devoted 10% of the effort to doing the physics and math that you do to handwaving and weasling, you would understand the science. Why do you think you have that huge hole at ~15 microns in the transmission spectrum of it’s not from CO2?
Remember it’s climate CHANGE. Yes, there’s more H2O than CO2, but it’s the CO2 that is increasing. H2O is changing only as we’d expect it to based on the rising temperature.
DO THE FRICKING MATH!
Dear Ray Ladbury RE: 305.
Thank you for that very thought provoking post.
I will bold your comment for visual clarity. Do not know the accepted convention.
1)assertion that CO2 does not have sufficient heat capacity to warm Earth (heat capacity is irrelevant to the radiative physics involved)
As I have come to understand radiative physics and said definition by realclimate.org convention or experts, your observation is true.
And it is also quite obvious that CO2 must have some other property than radiative physics that causes it to drive global temperatures.
2)your assertion that heat loss of Earth an isolated system surrounded by vacuum is dominated by convection
My assertion is that heat loss from the Earth comes from radiation. But the delay of the heat captured, the overall rate of cooling
involves convection in transporting said heat to an altitude where it escapes by radiation. And if you read my posts, I think I have explained
this several times. But as a picture is worth a thousand words. Nasa did a nice job creating an extremely simple view of the
mind boggling complex Earth greenhouse effect. So I repeat the link. http://e6.ath.cx/gw/2007.nasa.gov.energyballance.gif
3)You have taken data out of context e.g. using the 1998 El Nino as a peak of warming
and on and on.
Worst of all, Tom, theres no evidence your learning curve has a positive slope. Until you make an effort to learn the real science so that you can talk apples with apples, you are wasting our time and yours.
I don’t know what context I’ve taken any data out of. And I may have no idea what an apple is in your world.
I can only guess what data you are referring. I am guessing this is the bad apple. http://acute.ath.cx/BULK/global_warming/2008icecap.us-MonthlyCO2vsTemps.jpg
If I have guessed correctly, could you explain why it is out of context. Well out of what context.
There is a context of a real world following the laws of physics. In that world this is how I view chart above.
What do I see when I see this chart and read the explanation in the article where I found it.
The data graphed represents a total in some manner of the “temperature of the Earth” The temperature reflects the rms value of
the heat energy of the Earth. Is my supposition correct? When the rms value of the heat energy of the Earth goes up, the global temperature goes up.
The context for that chart is this. http://icecap.us/index.php/go/joes-blog/correlation_last_decade_and_this_century_between_co2_and_global_temperature/
The key point or context is that clearly for the last decade there is not a build up of stored energy. And nobodies CO2 radiation physics predicts this.
Tom, what molecules radiate the infrared photons that are most likely to actually leave the planet, and why?
Spencer Weart’s website explains this clearly.
One last try.
Tom: Yes, H2O and CO2 overlap in the lower troposphere, and thus H2O overwhelms CO2 in the lower troposphere, but H2O drops off sharply as you approach the mid-troposphere, while CO2 remains well-mixed to the top of the troposphere, and into the lower stratosphere. Thus it is here, above the point where H20 dominates, above the point where H2O drops off, that CO2 is free to absorb in the wavelengths that overlap with H2O lower down. And it is here that adding more CO2 to the atmosphere will make a difference.
As Ray wrote, we aren’t adding more H2O to the atmosphere, we couldn’t even if we wanted to, but we are adding more CO2. And CH4. And NOx. And CFCs, etc.
Think about it.
Dear Hank Roberts re: 311. A google of Spencer Weart’s website finds a person interested in the the history of physics. So do you have a link to a page you feel actually deals with scientific explanations and not history.
I believe just because a guy into history, he may have some thought or idea that makes me see differently or more clearly what I know.
But this google hit, #4, that also mentions Global Warming, does not recommend.
This was written as an evaluation of one or Spencer Weart’s books.
Weart’s presentation changes from a dispassionate history of science into a polemic, arguing that global warming is a human-caused reality, and implying that everybody who says otherwise is a shill of the coal and oil industries.
http://www.21stcenturysciencetech.com/Articles%202004/Spring2004/global.html
[Response: or part of a LaRouchite cult. Take your pick. – gavin]
Ray (308), thanks. One clarification (that brings back memories of an earlier excited discussion): I thought temperature (as we know, love and measure it) derives from only the three degrees of freedom in the translation (whole molecular velocity) energy pool. And not from the intramolecular energy pools of rotation and vibration degrees of freedom — in the first order. True? Or not?
Ray, tom: I’m surprised the NASA energy balance graph that tom referenced glosses over the gross upward IR radiation leaving the surface (114% per IPCC, e.g.) and the returning IR back radiation. Any thoughts?
Dear Jim Eager re; 312, Terms like lower troposphere do have meaning, but I like using altitudes. Anyway I will use this chart as my definition of altitude zones.
http://e6.ath.cx/gw/temp_vert-climateaudit.org.gif
Mid troposphere is 5 Km Temperature is -20C and pressure is about 1/2 an atmosphere. I believe, I interpret that the laws of physics mean that CO2 will not trap any serious amount of heat. I can only offer these intuative arguments. All black body radiation goes in a random direction. But all that go up and are of a wavelength that can be adsorbed go further than those that go down. Air is less dense going up and thus extinction distance is further. All going down also must pass through lots more absorbing molecules. The cooling of a dry desert at night is in degrees per hour. The dryer less dense air at 5K would likely cool at even quicker rates as that air has even lesser density thus lesser specific heat capacity.
Now also, If what you say is true then one would see the heating of that area. And by the way, http://e6.ath.cx/gw/IPCC-predicted-v-measured.jpg the IPCC in the last report published this figure showing exactly what you have described.
http://e6.ath.cx/gw/IPCC-predicted-v-measured.jpg
Now this is not gospel, but I don’t see how one can reconcile CO2 heating the mid to upper troposphere. And there is a mystery -70C spot in the middle of the Gulf of Mexico that is a fountain of cold.
Another interpretation of images I collected and studied. 2 weeks of GOES-East, Eastern North America IR(infra-red: 10.7 µm): Satelite a tcsh script build-global-IR-movie.com was run for a two week period and collected the images updated on the half hour. 688 images were collected. http://e6.ath.cx/gw/movie/test3/
This is a link to one image and there large areas of temperature above the clouds at far below the -20 reported as the 5KW temperature.
http://e6.ath.cx/gw/movie/test3/640×480/goes_enam-2008_01_28_12_09_IR_Large.jpg
Dear Ray Ladbury RE: 309
I guess I don’t know where to find the physics and math you are referring too. Is the absorption hole in a spectrum a clear indication of the idea of an extinction distance. That a wavelength is absorbed, the energy redistributed to surrounding molecules and even when they re-radiate their small amount of that wavelength, it gets absorbed immediately over and over?? With each re-radiation some goes to space and over time air cools as heat is escaping???
I would love to find the math and physics that shows this is not the case.
Ray in engineering there is an idea that when two things are in play and one is varying by orders of magnitude on numerous time scales and one is increasing by a fraction of a fraction, All thing being equal, a fraction of a fraction adds zero.
Tom,
Spencer Weart is also a physicist as well as a historian of physics. I know him personally and can assure you he is well conversant with the physics. Besides, the historical perspective is important, as it illustrates the blind alleys of climate research in the past (and down which you are stumbling at present). For something more technical, might I suggest raypierre’s text on atmospheric physics–a good starting point. There is also a workbook of problems–don’t skip them or you won’t truly understand the material.
Tom, one indication that you don’t know what you are talking about is the fact that you jump around from cause to cause to problem to problem and then start cycling through again. You have to look at things systematically. And you will have to unlearn the incorrect interpretation you have amassed through lack of systematic study.
As Mark Twain said: “What gets us into trouble is not what we don’t know. It’s what we know for sure that just ain’t so.”
A case in point is your vague reference to extinction. If you bothered to read the realclimate article “A Saturated Gassy Argument,” you would know that the effect of CO2 doesn’t saturate–in merely increases logarithmically. As the concentration of CO2 increases you start to get more absorption into the tails of the absorption band–and the tails of the band are lorentzianm, and so, thick.
BTW, although I am a physicist, the physics of my day job is very applied–making sure microelectronics will work in a high-radiation environment. One thing you should have learned is that when you have competing effects, a small change in one can drastically tip the balance of the entire system.
Rod,
Temperature is the partial derivative of energy wrt entropy. Now remember back to thermo in the dark and distant past. As you heat a gas, initially, the only degrees of freedom are those associated with kinetic energy of the molecule as a whole, so E~1.5*kT. However as you heat things up more, you start to excite vibrational, rotational and other internal degrees of freedom, and for each new mode, the proportionality between E and kt increases by 0.5. So if you have 2 rotational degrees of freedom, E~2.5*kT. Throw in a vibrational mode and you get 3kT. In other words, it takes more energy to increase the temperature as the temperature gets higher. The Wikipedia articles below aren’t bad references:
http://en.wikipedia.org/wiki/Specific_heat
http://en.wikipedia.org/wiki/Equipartition_of_Energy
Re #306
“This 1 to 5 relationship ignores the reality of 10 to 50 fold more H20 molecules and that H20 is a
total and partial absorber of far more wavelengths than CO2, and also H20 is a partial absorber of the far fewer CO2 wavelengths that are
in the black body radiation spectrums of the earth’s temperatures.
I have spent much time looking for and when possible annotating images to emphasize what seems obvious to me.
http://e6.ath.cx/gw/Atmospheric_Transmission-sm.jpg
http://e6.ath.cx/gw/Global_Warming_Not_From_CO2-fig1-n-2.jpg
Are all the equations of the models that compute CO2 driving temperature using the above weightings?”
In short, no.
You’re relying on inaccurate cartoons for your spectroscopic data which give a totally false picture. Water does not overlap with CO2 to the degree shown, neither spectrum is a continuum as shown but clusters of discrete lines which when viewed at high resolution do not directly overlap. Consequently in order to determine the actual absorption a detailed line-by-line calculation must be done, you can find the data used for this in Hitran: http://cfa-www.harvard.edu/hitran/
To get a better picture you can run calculations on-line using Modtran: http://geosci.uchicago.edu/~archer/cgimodels/radiation.html
This will give you a much more realistic picture of the atmosphere than the nonsense you have in those links above.
Tom, Larouche. Look it up.
Re #319
Ray just a minor correction, each vibrational mode gives two degrees of freedom so it adds kT not 0.5kT.
Dear Ray Ladbury re: 318 It is nice to hear you know Spencer Weart personally, And your being so familiar, I understand why you do not provide a link to a page of Spencer that shows his clear explanations so I can undo all my bad.
As you have proffered. you will have to unlearn the incorrect interpretation you have amassed through lack of systematic study.
I must unlearn, if no heat energy can be found, it is still there waiting to raise the temperature.
Dear Phil. Felton re: 320
As I read your comment, I smiled and asked, it the man hockey stick figure a cartoon. I have searched long and wide for cartoons that plot the absorption of CO2 and H20. Most are very cartoon like. They have no info really speaking to the metrology of how to quantify what is plotted.
Even NIST does not really do it. I do wonder why it is so hard. And I would be thankfull for any url pointers to accurate cartoons, of graphs or figures. Especially those the also list the complete math behind the figure, the assumptions etc.
This is a composite of NIST plots of CO2 and H20. I do not see the non overlap you are claiming. Also if one takes the NIST to have
some unit correlation. Yes I know it is not stated. Anyway, in that at and in the first hundred feet of atmosphere there are 10 to 50 molecules of H20 to CO2, A 5% H20 absorption is the same as a 50% to 100% CO2 absorption.
http://e6.ath.cx/gw/nist.gov-H20-n-CO2-trans.gif The NIST chemistry WebBook allow plotting with different scaling and units.
If you look at my raw directory http://e6.ath.cx/gw the separate frames used to make my cartoon are there. They are files the begin with nist-
Then I use http://www.spectralcalc.com/spectralcalc.php there black body calculator to create graphs of different temps.
Then one matches the scaling and one can apply them together making what you call inaccurate cartoons
In engineering, the use of graphical or cartoon methods is quite normal and regulars. So what rule of mathematics says using graphical methods to join cartoons is not the same as doing a detailed line-by-line calculation
Tom, would a language other than English be easier? Do you have a local library where you can get some of the references suggested? The footnotes in the AIP History would be easy for a library reference desk to locate for you and would be helpful to read.
Your website’s in .CX — Christmas Island. What sort of local help do you have available? Any science library or college nearby?
This seems as good a place to put this as any. Looks like an interesting upcoming PRL:
http://www.aip.org/pnu/2008/split/862-1.html
Tom Watson, Just what the hell do you think IR radiation is if it is not heat energy? I am afraid I don’t have much sympathy if you are unwilling to read the resources provided. I am afraid I don’t know how to surgically install understanding. Certainly you will not arrive at it solely by posting on a website. In my case, understanding is the product of concentrated effort. Why do you think it should be easy for you?
Ray (319), here’s my point, which one has to set aside for the moment the quantum mechanics which determines the probability of what energy goes to which degree of freedom, but what I think is instructionally accurate. Say E1 is applied at a low enough temperature that all of this energy (or less) goes into translation (1/2mv^2) so that the temp is (2)(E1/3k, and the temp changes linearly as E1 decreases. (I’ve picked E1 as a discrete shift point.) Now add a bunch of energy E2 = deltaE + E1 so that all of deltaE goes into, say, rotation, so that E2 = 5kT/2. You now end up with 66% more energy in the molecule with zero change in the temperature.
Being a little more realistic, say that E1 still is all translation but deltaE gets distributed between translation and rotation according to an approximation of quantum distribution and equipartition. I contend that the portion of deltaE that goes to translation increases the measurable temperature, that going to rotational does NOT. Likewise when radiation is absorbed (always either rotation or vibration) the temperature, in the first order, does not change Correct or not?
Dear Hank, thank you, I did not know .CX was associated with Christmas Island.
But a whois ath.cx is more informative.
…Registrant:
DynDNS Hostmaster
Dynamic Network Services, Inc.
1230 Elm St.
5th Floor
Manchester, NH 03103
And [ 247 ] > host e6.ath.cx
e6.ath.cx has address 76.254.193.1
and [ 248 ] > host 76.254.193.1
1.193.254.76.in-addr.arpa domain name pointer adsl-76-254-193-1.dsl.mrdnct.sbcglobal.net.
I have a dyndns account, but it was free for 5 domain as I recall. dyndns and several other services allow for a domain behind a dhcp gated router to have a constant URL.
And I would welcome any suggestions to references you can provide. What you show me, you show the world of your expertise and knowledge. Do you prefer paper or the internet as a library? And I have a photo gallery for all who enjoy a pig out on reading…. http://toms.homeip.net/2003.07.08_pig_out_on_reading/
As embarrassing as this is to admit, I’ve scanned all four 4th assessments and can’t find the IPCC Figure 1.1 above. Can somebody tell me where it is to be found? I need it for an ongoing [sigh] debate about AGW with an energy industry engineer. (BTW, you guy’s efforts have been INVALUABLE!)
[Response: Chapter 1, page 98 in WG1 report. – gavin]
Here’s another unhappy effect to model:
Rod,
It’s not quite as simple as that. Temperature is not defined for a single molecule, really, so what you will have is a distribution. At low temperature, a very few molecules will have high enough energy that they can excite rotational and vibrational modes in themselves and other molecules via collision. However, the vast majority will only respond via changes in translation. Now raise the temperature–more and more molecules will have high enough kinetic energies to respond rotationally or vibrationally. Now suddenly, you will have a much greater proportion of the energy going into these modes, so the specific heat will rise (i.e. it will take more energy to cause the same delta T).
You will just drive yourself crazy if you try to think of temperature as being a property of a single molecule. Temperature is what they call an intensive property, as opposed to extensive properties like energy.
Ray (331), you say, “…Now suddenly, you will have a much greater proportion of the energy going into these modes [rotation or vibration], so the specific heat will rise (i.e. it will take more energy to cause the same delta T). This means the energy raising the specific heat by parking in rotation and/or vibration modes, prima facie, does not raise the temperature. Correct.
We all went round and round on the following. I don’t want to restir the pot, but, for the record, I have no discomfort at all with the concept of a single molecule having temperature. You say it takes a bunch. I say how many? You say 100,000 at least. I say, O.K., how about 50,000? ….ad nauseum until I say, how about 4? …. You know how this works: you can not support eventually saying NO at any discrete number. While it is true that their is an energy distyribution within a gas such that there is an average 1/2mv^2 that determines the [average..] temp. None-the-less each individual molecule in the gas is zipping around at some discrete velocity with a discrete energy level (though probably none exactly on the average) and experiencing a discrete temperature in Kelvin — even if we can not measure its temp. I recognize temperature defined by change in entropy presents big problems here, but this is not the only dichotomy in physics we have to live with (wave-particle anyone?); besides I think the formula is defining entropy in term of temperature (as one ind. variable), not vice versa.
ps I said I didn’t want to stir the pot, …then went and did! Plus my proofreading was atrocious. Sorry
http://www.aip.org/history/climate/Radmath.htm
http://www.aip.org/history/climate/links.htm
Guys, I think we should stop responding to Tom Watson. For whatever reason, he is either not reading the stuff we refer him to or is not understanding what he does read, and there’s nothing we can do about that. Trying to educate someone who either cannot be educated or, more likely, doesn’t want to make the effort, is a waste of time.
[Response: Chapter 1, page 98 in WG1 report. – gavin]
I knew it was going to be embarrassing. Thanks.
[edit]
Dear Barton Paul Levenson re 335: What links do you proffer I have been given and not read and not understood. What links have you given that you claim I have not read and not understood.
Do you think it took no time to compose all the posts I have made on this forum.
Do you think it took no time to compile all the info on my open web directory and to link to specific figure located there. Do you think it took no time, not effort no understanding to edit and annotate and try to combine information so it makes more sense.
I have posted here with honest questions. My questions about things that don’t add up. is there any point in saying more about you post???
Dear Ray Ladbury RE 326
I have started looking at http://www.aip.org/pnu/2008/split/862-1.html, I posted a reply just before this to BPL.
The nature of how this forum loads in my browser is such I see the last posts before I see the time continuation of where I left off
reading some previous time. So when I wrote the last post, I had not read 326. I confess, after spending time at
wiki.
http://en.wikipedia.org/wiki/Beer-Lambert_law to
http://en.wikipedia.org/wiki/Absorbance to
http://en.wikipedia.org/wiki/Absorption_spectrum
I do not really know what IR radiation is? smiling here… yes IR radiation is energy, how it’s absorbed, I hope to better understand.
I know what I think IR radiation is. But I am searching to find out if what I think is correct or hogwash.
Now I have spent time composing this reply, not reading the link you provided and the links it refers too. I now post this and start looking at the link provided.
And thank you for that link.
By the way, I was at wiki attempting to make heads out of your earlier posts on molecular vibration. This is not a knock. Different folks speak with different jargon. I could only guess as what you were saying.
Tom Watson, If you have specific questions about what I’ve said about vibrational modes or anything else, I’ll try to clarify them. IR radiation is simply electromagnetic radiation (photons) with wavelengths longer than visible light and shorter than microwaves. We feel it as heat, when it is incident on us, because it excites vibrational and rotational modes in the molecules in our skin (think heat lamp).
Tom, a suggestion. A comment forum like this does not lend itself to a thorough education. It works best for you to find some good resources–e.g. raypierre’s climate book–read them on your own and if you have specific questions, ask them. Provided they don’t hijack the discussion and aren’t grossly off-topic, you’ll usually get a courteous response. Look, climate science is an interdisciplinary study. It involves lots of different material and takes time to learn.
Rod,
Remember that in the expression E=1.5kT, the E is actually E-bar, the average energy of the ensemble. You will always have energies much higher and much lower, and the distribution will be roughly Maxwellian. Thus even at cold temperatures high in the atmosphere, there will be some collisions that excite CO2 atoms into their excited vibrational state. As to the answer of how many molecules one must have to have an ensemble, the answer is how large an error can you deal with on your temperature, pressure,… With as few as 100 molecules, average errors will be ~10%, so nanoclusters do have a temperature.
Dear Ray Ladbury RE 326
I have now looked at http://www.aip.org/pnu/2008/split/862-1.html, Overall it reveal little to nothing new to me.
It says A new joint study by French and Russian scientists shows in detail how carbon dioxide molecules absorb and sometimes scatter light energy not only singly but also during inter-molecular collisions but reveal no netails.
It does reference http://www.aip.org/png/2008/300.htm as this have now derived the first exact mathematical formulas that can be used to calculate how collisions between molecules modify the absorption spectra for those molecules (see figure at
But looking at …300.htm is a statement of what most may believe, but nothing about anything in formulas.
I do believe, but will not claim with certainty that I have read previously various pages at aip.org. It general the pages seem not very definitive.
I do have an undisciplined method of saving links I encounter in a file http://e6.ath.cx/gw/aaa-goodlinks I also now more regularly use
an open source program, the simple xpaint to annotate figures I download with the original link for future reference.
Now back to the discussion of absorption, http://en.wikipedia.org/wiki/Absorption_spectrum There is a claim, or explanation and it does note a cite is needed. But this explanation uses an example where we have parallel traveling photons from the emission source.
in both the atomic and molecular cases, the excited states do not persist: after some random amount of time, the atoms and molecules revert back to their original, lower energy state. In atoms, the excited electron returns to a lower orbital, emitting a photon. In molecules, the vibrational or rotational mode decays, also emitting a photon.
When this decay occurs, the photon produced is not necessarily emitted in the same direction as the original photon. The most common angle of this has been shown to be about 45 degrees of the original photon[citation needed]. This applies to any situation where gases lie between a light source and an observer: the observer will see gaps in the spectrum of the light corresponding to the wavelengths of the photons which were absorbed. These gaps occur despite the re-emission of photons because the re-emitted photons are equally likely to travel in all directions, and it is statistically unlikely to travel along the original path to the observer. These gaps appear as black lines in an image of the spectrum.
But width of the earth emitting (let’s says by hundreds of square miles) and the thinness of the atmosphere. (choose some altitude) has the IR wavelength being emitted at all angles with respect to an observer at some altitude.
If one sees absorption lines of a specific band at some altitude, the wiki 45 degree and re-emission explanation seems incorrect to me. It seems absorption with conversion to heat and then re-emission of full spectrum where same absorbed band but less energy gets re-absorbed and band shifted component of energy now or will recursively contain all energy in the original absorbed band.
That part of the energy that is not absorbed leaves the system. That energy that is absorbed remains and is constantly part of the heat within the earths system.
But I still have found no explanation of how C02 has any unique property the accounts the supposed change of some PPM.
Tom, First, the cite I gave from physics update was not intended to be tutorial. It is a result that will be published soon in Physical Review Letters. It is probably beyond your current understanding, but even if it were not I would not expect you to understand it based solely on a news release.
One thing from the article should give you a hint though–it said at sea level a CO2 molecule undergoes ~10^10 collisions per second. Add to that the fact that the excited vibrational state of CO2 lasts of order microseconds, and it should become clear that relaxation via collision is much more likely than relaxation via photon emission. The energy is thus shared mainly with N2 and O2 which (except in extreme conditions) do not emit in the infrared. The whole atmosphere heats up–it’s not just that the CO2 slows down the escape of the IR.
Dear Ray Ladbury RE: 339,
I had not read 339 yet when I posted my reply #341. I don’t know that I need mention that for others, but I need to note it to have the dialog make sense to me if I refer back to it in he future.
Your observations about the forum are valid. As to vibrational modes, I have read many different renditions of word descriptions to create a minds eye visualization of the interaction of photons with matter and in particular what absorption is like.
I have reveiewed many text books, The specifics of the vibration modes are interesting, but have not addressed a quantification of probabilities of what happens when those photons that are tuned to be adsorbed by a specific molecule or molecules in general do intercept.
The state of wisdom is as the link you referenced in #326 http://www.aip.org/pnu/2008/split/862-1.html The state of knowledge seems to be this.
and its ability to absorb and trap infrared radiation is thought to be instrumental in producing greenhouse effects. CO2 molecules one at a time can absorb light. But molecules can also absorb light when they collide with other molecules. This collision-induced absorption, occurring at wavelengths different from those for single molecules and accounting for about 10% of overall IR absorption, is insufficiently understood.
So if this belief of mine is correct, across realclimate.org there is an attempt to lower the “overall IR absorption, is insufficiently understood.” I jumped in and asked what I judged to be questions that go to the center of ambiguity in understanding.
I have no wish to hijack a forum, But one can say my posts have. But I do provide links to web resources I control and from them who I am and where I live and how to contact me can be found, even by non rocket scientists. I am open to any suggestions on how to follow my curiosity on how it works.
So thank you again for taking the time to post responses to my concerns, questions and ramblings.
How many of those who post, can you look in the eye???? http://acute.ath.cx/watson/pooter/ this is dated 04/06/99 so the eyes are older now.
PS as I posted the preview this time I checked an saw 342. So Re 342, I would not assume you would not understand an explanation of anything by someone who actually understood it. I find that rule is useful in general.
I find I ofter understand better for having explained to others who did not understand. I have also witnessed arguments between folks saying the same thing.
As to the 10^10 of collision and usec atomic level heat conduction times, I would guess photon time of flight is sub picosecods. Does this suggest the more CO2 more heat faster transfered to air, Does this agree with the idea of a shorter adsorption distance. More molecules, sooner intercept. Does that imply a more concentrated initial heating near surface that means sooner of faster start of convection of warmer less dense air upward.
But H20 is still 10 to 50 times CO2 in molecule population.
> photon time of flight is sub picoseconds
“mean free path”
> adsorption
Wrong word, wrong concept
> But H20 …. molecule population
Altitude: “Water is down to CO2 levels by 6km”
http://rabett.blogspot.com/2008/01/if-you-dont-remember-past-you-will.html
First, H20 isn’t increasing except as a function of increasing tempeature–remember it’s climate CHANGE. Look for what’s changing. I meant no offense when I said you probably would not understand the research linked at APS. It’s state of the art research, and you don’t have the PRL paper to reference.
IR absorption–in band–is quite well understood. It is a question of how that absorption gets enhanced as a result of intermolecular collisions. This is one of the reasons why CO2 absorption doesn’t saturate.
The speed of a photon is irrelevant. What matters is how many CO2 molecules it will encounter on its way out of the atmosphere. Each CO2 molecule increases the probability of absorption and the energy trapped. And keep in mind that excited CO2 can radiate at any level of the atmosphere and can be absorbed by CO2 at any level into the stratosphere. Really, check out Raypierre’s book. It will help.
Ray, your implication will come as a surprise to most 101 textbooks,… and to my buddy Feynman, which use a single molecule to start the development of the 1/2mv^2 = 1.5kT stuff.
It’s E-bar because that’s what we choose — as the only useful quantity to work with. But you’ll have a long row to hoe to convince me a lonely singular molecule has no kinetic energy. Possibly our disagreement stems from the fact that my single molecule with K.E. and a Temp is interesting in concept but does no physicist any practical good, other than theory: One can not (accurately?) measure either its temp or its velocity. Measurement can have 100% error. But that does not mean its velocity and temp does not exist.
Isn’t IR absorbed one molecule at a time (granted with teensy succession times)?
I was reviewing the forum. I found, I spent so much time addressing less than flatering replies to my posts and considering how to reply in way both informative and civil. that I missed other posts and posters that I believe I share very common state of thought.
I continue to post as I am still attempting to expand my understanding. It may be a waste my time if no one reads or no one replies, but my efforts of attempting to write down what I believe and explain it helps me better understand what I believe and often furthers my own understanding.
Dear Gene Hawkridge RE 27 and a general comment to all.
Your said “That is clearly not healthy are the nuts who conclude, based on a few years data, that global warming is fictional ”
My skeptism is that any link between a variation of PPM of C02 and an ability to drive climate anywhere near the current IPCC supposed levels has ever been explained beyond a supposition and nothing near proven or even likely. It is all suppositions, and theories.
I posted on this tread hoping those who freely volunteer the time to create such an extensive site and state
RealClimate is a commentary site on climate science by working climate scientists for the interested public and journalists. We aim to provide a quick response to developing stories and provide the context sometimes missing in mainstream commentary. The discussion here is restricted to scientific topics and will not get involved in any political or economic implications of the science. https://www.realclimate.org/index.php/archives/2004/12/about/
would be a place where I could ask questions or explain why I am confused about C02 suppositions of massive impact.
I hoped that such learned and expert folks would be able to explain in laymans terms the nature of exactly how CO2 is supposed to drive temperature. But Laymans terms using some quantification that explains how mass in part per million has some effect in parts per ten or hundred or even a thousands.
I have extensive experience in using all manner of sensors and instruments to measure physical properties and then using various engineering math to process, create simple visualizations. Visualization that greatly assist any laymen in grasping the complexity in a real world system.
I attempt to express in an EE jargon.
In electronic systems there is signal and noise and a signal to noise ratio. How does the signal of ppm of CO2 drive the signal of Temperature when H20 is so much higher a signal in PPM, Multiplied by higher absorber of power of the spectrum of interest.
In the Earth system
The water signal has always been present, the water signal is orders of magnitude larger and varying in amplitude by orders of magnitude over the current small PPM increase signal of CO2.
Also heat by conduction and convection and radiation get to a place where it escapes via radiation. The massive for all time present signal of H20 also can also tranport via convection about 300 time the heat of CO2, O2, and N2, per unit mass. 02 and N2 make up 96 to 98%. Water makes up 1% to 3%. So does this mean that in a cubic foot of air that H20 by convection can transport 3 to 9 times the heat in its 1 to 3% as much as the entire 97 to 99% of the rest of the air that includes some PPM of CO2.
300 tims per unit mass.(specific heat, heat of fusion, heat of vaporization and only h20 is going through phase changes in atmospheric convection.)
I always attempt to parse what a skeptic is skeptical about. I don’t get how one can add the H20 and CO2 signals and support the CO2 impact suppositions.
For my efforts and questions, this is a summary of what has
The collective argument is that one must read more to understand and those who cannot are just of too limited an intellect to comprehend.
Dear Hank Roberts Re: 344.
I would suggest “photon time of flight is sub picoseconds” is good layman terminology for the technical “mean free path” I often use the best layman terminology I can think of, even if I did know the appropriate technical jargon. But such corrections are welcome as then all may learn appropriate technical jargon.
adsorption vs absorption is a db typo. My typing sucks. I also often put you instead of your and the where this and that should be. I omit the letter s often and may include it wrongly. That is a handicap I have.
But on adsorption vs absorption, I wonder if as I think constantly in pictures and mixed concepts, is the electric field of electrons in obit that is selective about which photons may enter functioning like a membrane. Does my many levels mental processing somehow impact what signal gets to what finger to execute a particular character.
I know my minds eye is beyond the speed of my thought to finger communications.
I can only say sorry for pain it causes. It is more work to listen to garbled speech. I have tried many schemes and still my mind in review does not catch all my garbles.
Beware “word salad” — picking terms from other areas and tossing them into postings about climate make it harder to find relevant research.
E.g., “photon time of flight” — this is from biomedical laser work and will find articles on seeing through flesh not through atmosphere.
Problem between chair and keyboard is common enough; drafting offline is always useful to allow time to edit til comprehensible by others.
Else people just skip when they expect to be baffled. Enough said.
Re #347
“In electronic systems there is signal and noise and a signal to noise ratio. How does the signal of ppm of CO2 drive the signal of Temperature when H20 is so much higher a signal in PPM, Multiplied by higher absorber of power of the spectrum of interest.”
Firstly, the concentration isn’t the signal and secondly haven’t you ever heard of gain?
Did you ever take a look at MODTRAN as I suggested?
Re #324
“This is a composite of NIST plots of CO2 and H20. I do not see the non overlap you are claiming.”
Several reasons for that, firstly you superimposed graphs of a known concentration of CO2 with an unknown (but high) concentration of H2O, secondly you plotted the graph over too large a range of cm-1 (no point in anything over 2000 cm-1), thirdly in order to see the overlap you have to plot at higher resolution closer to the line-widths.