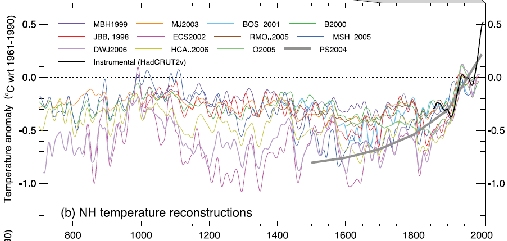
Much research effort over the past years has gone into reconstructing the temperature history of the last millennium and beyond. The new IPCC report compiles a dozen reconstructions for the temperature of the Northern Hemisphere (including of course the original “hockey stick” reconstruction, despite opposite claims by the Wall Street Journal). Lack of data does not permit robust reconstructions for the Southern Hemisphere. Without exception, the reconstructions show that Northern Hemisphere temperatures are now higher than at any time during the past 1,000 years (Figure 1), confirming and strengthening the conclusions drawn in the previous IPCC report of 2001.
Fig. 1: Figure 6.10 (panel b) from the paleoclimate chapter of the current IPCC report (see there for details).
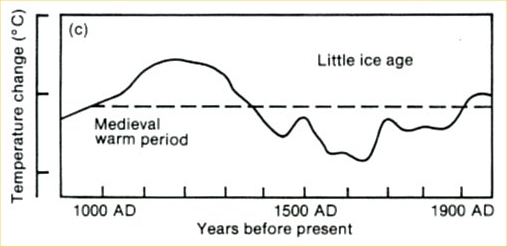
“Climate sceptics” do not like this and keep coming up with their own temperature histories. One of the weirdest has been circulated for years by German high-school teacher E.G. Beck (notorious for his equally weird CO2 curve). This history shows a medieval warm phase that is warmer than current climate by more than 1 ºC (see Figure 2). So how did Beck get this curve?
Fig. 2, modified from E.G. Beck (we added the green parts).
The curve is a fake in several respects. It originally is taken from the first IPCC report of 1990: a scan of the original is shown in Figure 3. At that time, no large-scale temperature reconstructions were available yet. To give an indication of past climate variability, the report showed Lamb’s Central England estimate. (Unfortunately this was not stated in the report – an oversight which shows that IPCC review procedures in the early days were not what they are now. We will post in more detail on the history of this curve another time.)
Fig. 3. The past millennium as shown in the first IPCC report of 1990, before quantitative large-scale reconstructions were available. This curve was based on Lamb’s estimated climate history for central England.
But Beck did not stop at simply using this outdated curve, he modified it as highlighted in green in Figure 2. First, he added a wrong temperature scale – the tick marks in the old IPCC report represent 1 ºC, so Beck’s claimed range of 5 ºC exaggerates the past temperature variations by more than a factor of three. Second, the original curve only goes up to the 1970’s. Since then, Northern Hemisphere temperatures have increased by about 0.6 ºC and those in central England even more – so whatever you take this curve for, if it were continued to present, the current temperature would be above the Medieval level, as in the proper reconstructions available today. As this would destroy his message, Beck applied another fakery: he extended the curve flat up to the year 2000, thereby denying the measured warming since the 1970s. With this trick, his curve looks as if it was warmer in Medieval times than now.
When approached directly about these issues, Beck published a modified curve on a website. He changed the temperature range from 5 ºC to 4.5 ºC – but he shortened the arrow as well, so this was just cosmetics. He also added instrumental temperatures for the 20th Century at the end – but with his wrong temperature scale, they are completely out of proportion. (In fact his version suggests temperatures have warmed by 2 ºC since 1900, more than twice of what is actually observed!)
Beck goes even further: in a recent article (in German), he has the audacity to claim that his manipulated curve is right and the more recent scientific results shown by IPCC are wrong. And for years, he has offered his curve on an internet site (biokurs.de) that distributes teaching materials for schools, with support from German school authorities. It is quite likely that his fake curve has been shown (and will continue to be shown) to many school children.

I just wanted to thank you for an excellent site. I am somewhat skeptical to the ‘climate-change story’. However, being a scientist in an unrelated field, I am painfully aware of how little I know about the science behind ‘the story’.
This site helps a lot by presenting the science both in sufficient detail and in an accessible way. The fact that you also seriously address counter-arguments on the net is a very important feature of your work.
If anyone are to rid me of ‘climate skepticism’ it will be realclimate.org :-).
DocMartyn, could you please understand that the atmosphere is not in steady state? The increase of 1 to 2 ppm per year disproves the steady state assumption.
The natural carbon influx is NOT 21 GtC. In fact, the net effect of nature is actually a carbon sink, not an influx, on the order of 3 GtC.
The “lifetime” of CO2 is NOT 9 years. While it is often approximated as 100 years, I believe that William pointed you at a Realclimate discussion on the limitations of assigning CO2 a lifetime at all.
The reason that C14 disappears quickly from the atmosphere is that there are large balanced fluxes into and out of the ocean and into and out of the ecosystem (look up net ecosystem production, net primary production, net gross production – NEP, NPP, NGP to see different orders of magnitude for these)
And finally, the difference between spring and autumn is going to be much more an effect of vegetation growth and decay than any sea surface temperature effect.
I suggest you read http://ipcc-wg1.ucar.edu/wg1/Report/AR4WG1_Pub_Ch07.pdf the IPCC chapter on geochemistry and the carbon cycle in particular.
DocMartyn (#242) wrote:
The biggest problem with this is the seasonal variation itself. Plant exhalation is during the winter and peaks in April, just as the weather is beginning to warm up. The inhalation maxes around October. This is entirely out of phase with the effect you wish to measure. However, even if it were in phase, it would swamp the effect you wish to measure. Eliminate it, and you still have the seasonal effects of human emissions – which you would still have to factor out.
This shows the seasonal variations at Mauna Loa…
NOAA In Situ Carbon Dioxide (CO2) Measurements
http://www.mlo.noaa.gov/programs/esrl/co2/co2.html
Atmospheric CO2 reaches a maximum in April, minimum in October – and the cause in plant respiration – where plants take on CO2 during the warm months and release CO2 during the cold months.
Dear Marcus Re #252,
“DocMartyn, could you please understand that the atmosphere is not in steady state? The increase of 1 to 2 ppm per year disproves the steady state assumption.”
It does not by any means, please not that the seasonal variation in CO2 in greater than 4 ppm. It is therefore quite reasonable to treat the system as a steady state.
“The natural carbon influx is NOT 21 GtC. In fact, the net effect of nature is actually a carbon sink, not an influx, on the order of 3 GtC.”
This argument means you have no understanding of kinetics. Imagine the atmosphere from 100 BC to 1900; according to the ice record the steady state concentration of CO2 in the atmosphere was approx. 280 ppm for 200 years. During all that time the amount of CO2 entering the atmosphere was the same as that exiting the atmosphere.
Are you by any chance confusing the term “natural”, which means without human interference and “Biotic” which means a process caused by the presence or activity of living things?
“The “lifetime” of CO2 is NOT 9 years. While it is often approximated as 100 years, I believe that William pointed you at a Realclimate discussion on the limitations of assigning CO2 a lifetime at all.”
So you and Willam are correct and the actual data is wrong? The summing of rate constants, based on guesstimates, is not a way to work this out.
Half-life is a real kinetic term, and does not only apply to first order rate kinetics, it is what it says, the average time it takes for 50% of something to disappear. We can talk about the half-life of CO2 or 14CO2 in the atmosphere and we can also measure it. I have shown two calculation which demonstrate that the half-life of 14CO2 and CO2 is between 9 and 12 years. I am prepared to read and debate other other data sets or other interpretations of the datasets already presented. However, I will not accept the argument that I am wrong becuase I am wrong and someone else is right.
“The reason that C14 disappears quickly from the atmosphere is that there are large balanced fluxes into and out of the ocean and into and out of the ecosystem (look up net ecosystem production, net primary production, net gross production – NEP, NPP, NGP to see different orders of magnitude for these)”
Why are fluxes “balanced”? Do CO2 molecules get issued with a map and a set of instructions everyday telling them where to go?
Actually, your first line is essentially correct for a system at steady state, the influx and efflux from the atmosphere is equal. The efflux is the sum of all processes that result in CO2 disappearing from the atmosphere and the influx is the sum of all the input sources with respect to time. This is why we know that influx of 12CO2 and efflux of 14CO2 resulted in half the atmosphere being turnedover in a decade or so.
“And finally, the difference between spring and autumn is going to be much more an effect of vegetation growth and decay than any sea surface temperature effect.”
This is what is known in the trade as a postulate, it hasn’t even got as far as being a theory. This is a possible reason, but has not yet been established that it is the correct one. I suspect that the change may be due to the difference in the surface temperature of the Earths ocean/water surface at different times of the year and its productivity. So I would like to know, if possible, the monthly changes in water surface temperature and chlorophyll, world wide averged for the actual water surface.
“I suggest you read http://ipcc-wg1.ucar.edu/wg1/Report/AR4WG1_Pub_Ch07.pdf the IPCC chapter on geochemistry and the carbon cycle in particular.”
I think you will find that many people walked away from this simplistic box method of kinetic analysis in the late 70’s and then moved on to both steady state and flux control analysis. The reason for this was that adding up individual rate constants and assigning amplitudes to bits and pieces doesn’t actually work. Complicated systems are actually both simpler and and more complex than they seem.
Re 238: Perhaps I’m missing something here, but I find that the argument that CO2 is more abundant close to the surface and therefore its effectiveness as GHG is reduced does not make sense. First, is CO2 not considered as a well mixed gas for all practical purposes? Second, if the surface is the emitter of IR radiation, the most effective reflective layer will be close to the source. Higher, the reduced density and increased relative surface area would make it a loss less effective. I other words, if you want to keep heat with a blanket, you don’t suspend it a meter high from the body but instead you wrap it around. So I don’t see how having the CO2 more concentrated near the surface (if it actually is) makes it less effective as a GHG.
O.K. let me put it another way. Your bedroom is cold and so you put on a thermally insulated blanket, still cold so you add another blanket. At what stage to you need to get to before adding additional blankets will not have a great effect? Each blanket added to your bed only changes the heat transfer rate by a fraction or the heat loss.
So at the moment with 380 ppm CO2 no IR from the major CO2 absorbtion peals from the surface is tranfered above 10 meters. The only heat being trapped is from IR emissions from the atmosphere. If IR from the surface can’t move more than 10 meters up, why should IR generated at 11 meters be able to move down?
Philippe Chantreau (#256) wrote:
CO2 (and ozone) are responsible for the greenhouse effect as it takes place in the stratosphere – which is especially dry. Water vapor is responsible for the greenhouse effect as it takes place in the troposphere – near the surface.
Please see:
re: #251 Arne B
Maybe you can help out, given that the good folks who run this site like to provide info for people who will actually consider data. I’m very interested in why people think what they think about this, so perhaps you’d consider quick comments on the following:
1) How much time have you spent looking at AGW? and for how long? Have you been watching long enough to see which positions change (or don’t) as new data arrives?
2) If you’re skeptical (in the classic sense, not in the hardcore denialist sense), where are you on a 1-99% scale, i.e., if following the IPPC, virtually certain = 99% on the proposition:
“GW is happening, and the rise since 1950 is mostly caused by humans?”
I ask because when someone calls themselves a skeptic, it means different things:
80%: I think it’s real, but I still have a few reservations.
50%: more like “Not proven, but could very well be true”
and
1%: “I really don’t believe it, and it would take a lot of evidence”
3) Scientists tend to believe believe by default that strong scientific consensus in another domain is probably a good approximation, even while reserving judgement. So,if not:
a) Is this from “Haven’t had a chance to study in depth yet, and I never believe anything until I know more”?
b) Is it from irritation at some of the well-known “alarmist” exaggerations, especially from before the evidence had accumulated? Put another way, classic skeptics often see irrational/rational fights. Exposure to alarmism often pushes normal skeptics, not into the usual rational-skeptic (scientist) group, but into a 3rd group (denialists), where they stick via anchoring effects, plus the work that it takes to sort through the organized disinformation. 3-way fights are confusing to people used to 2-way ones, comparedto peopl who are not so normally skeptical, and just willing to take mainline science’s word.
4) Do you have a specific list of reservations/reasons to doubt and could you share those? and maybe which ones used to be reservations, and got crossed off? [For example, my list once had the disagreement between ground stations and satellites, but that got crossed off.]
5) Are there particular sources from which you derive these? Which ones do you like?
DocMartyn (#257) wrote:
Because… it gets re-emitted? Because… the atmosphere gets warmer and has its own thermal radiation?
The thermal radiation isn’t trapped at 10 meters or 20 meters or 1000 meters or whatever forever. The system ultimately rises in temperature to the point that the thermal radiation going out equals the thermal radiation going in, but with greenhouse gases absorbing the infrared and re-emitting it in both directions, the temperature has to go up for this to occur. This is the principle behind the greenhouse effect. It is the reason why the average temperature of the earth isn’t 0 degrees Fahrenheit.
It helps to understand a theory before you try and attack it.
PS
Thank you for providing me with a foil. I now consider the lesson complete.
Re: 257 DocMartyn said, “The only heat being trapped is from IR emissions from the atmosphere. If IR from the surface can’t move more than 10 meters up, why should IR generated at 11 meters be able to move down? ”
Now you are just being silly. You do realize that Zeno’s paradox was solved, right? Some IR does get back to the ground precisely for the same reason that some of it escapes to space–the photons are emitted isotropically each re-emission. You can do a Monte Carlo calculation and figure what fraction escapes and what fraction winds up reabsorbed by Earth.
In answer to your blanket analogy, Earth’s thermal insulation could certainly be more efficient, given that our neighbor toward the Sun finds itself at about 750 Kelvins, while we are at a pleasant 300 Kelvins. The greenhouse effect is good for about 30 degrees for Earth, while most of Venus’s temperature is due to its greenhouse effect (it only gets about 2 times as much solar radiation as Earth). We really do understand the greenhouse effect. Really.
DocMartyn: The fact that the seasonal variation in CO2 is larger than than the year-to-year change does not mean that a steady state approximation is reasonable. Note that the decadal increase is almost 20 ppm – that’s much larger than the 4 to 6 ppm yearly variation – does that help you understand what a non-steady-state system looks like?
Effectively, there are two reasons that the system is not in steady state:
1) Humans are dumping 7+ gigatons of carbon into the atmosphere/ocean/biosphere system. Given that the long term sinks (carbonate weathering and deposition of carbonates in the deep ocean) are less than half a gigaton, this alone would keep us out of any steady state.
2) The ocean has not had sufficient time to equilibrate with the new atmospheric concentration. If we were to magically hold CO2 concentrations in the atmosphere constant at 380 ppm, the ocean would keep absorbing carbon for centuries (the circulation of deep ocean currents is on the order of 1000 years, and there really aren’t any fast mixing processes between the upper ocean layers and the deep ocean)
William, I, the many authors of IPCC Chapter 7, the vast majority of scientists who work in the field, _and_ the data are all correct. You might want to take this as a hint that you should reconsider whether there might be a flaw in your method.
Fluxes are balanced (or almost balanced) precisely because for several thousand years before humans started burning fossil fuels the system was in equilibrium. Every tree that grows eventually dies. The ocean is in equilibrium with the atmosphere – every CO2 molecule that enters is eventually balanced by a CO2 molecule that leaves. These fluxes are LARGE in comparison to human emissions. However, humans are taking fossil fuels that were not part of this short term (eg less than geologic time) carbon cycle and moving more carbon into the atmosphere. This actually leads to a small imbalance in fluxes. The increased atmospheric concentration leads (because of basic laws of thermodynamics) to the ocean absorbing carbon from the atmosphere. It also leads (because of carbon fertilization effects) to the ecosystem also absorbing carbon from the atmosphere.
Because these fluxes are large, you can’t use the C14 tracer to do what you want it to do. Here’s a thought experiment: you have two jars. One has 20 white marbles (call it the ocean), one has 10 white marbles (call it the atmosphere). Every day, you take ten marbles out of each jar, and swap them. Now, add 1 black marble to the atmosphere jar. Now, when you do your swap, there is a 91% chance that the black marble will move to the ocean jar. By your logic, this means the half-life of carbon is much less than a day. But look! There are still 11 marbles in the atmosphere jar. From now on, by the rules of this game, there will _always_ be 11 marbles in the atmosphere jar. Therefore, even though marbles exchange rapidly between jars, the addition of a marble to one jar has effectively an infinite lifetime. Now, this isn’t _quite_ parallel to our situation: when we add marbles to the atmosphere jar, thermodynamics tells us that we actually increase the rate of marble movement to the ocean jar, until partial pressures balance again. But it is closer than your model.
Now, go read that IPCC chapter. The numbers they have are not derived using “guesstimates of rate constants”, but rather experimental data, using both bottom-up and top-down methods. People measure the carbonate in the ocean, they measure the carbon in soils and trees, and use these measurements to derive the carbon fluxes into the various components of the atmosphere/ecosystem/ocean systems. There is still uncertainty in the precise values: but there are two numbers which are fairly well known. That is the human fossil emissions (~7 GtC) and the increase of carbon in the atmosphere (4 GtC). The difference between these two numbers (3 GtC) MUST be the net carbon sink by the land/ocean system.
Also, on my postulate about ecosystems being the cause of the seasonal CO2 cycle: given that the spring/autumn cycle is much larger in the northern hemisphere (with large land masses) than in the southern (small landmass, large ocean), it makes much more sense to attribute it to ecosystems than to oceans. Plus, we can measure the CO2 emitted by leaf litter decay and show that it is of the right magnitude to cause this cycle.
Now, please go read the IPCC chapter and think about it. And look at your model, and think about where the carbon is going: if humans burn fossil fuels, and the carbon goes into the atmosphere, where does it go? It has to go into the ocean, or into plant matter, or it stays in the atmosphere.
Now, your little kinetic model has it magically disappear. You have human emissions, and you have natural emissions, and you have a half-life which basically says that carbon goes “poof”. But it doesn’t.
And now, I should go do something useful with my time…
[[isn’t it more likely that these photons, after being absorbed and emitted and reabsorbed and reemitted will simply escape into space, even if only because of the curvature of the atmosphere and the simple fact that there’s less air up there to absorb all those photons?]]
Certainly. The Earth will continue to radiate just as much energy as it absorbs from the sun. But how that energy is distributed while it’s in the Earth system can change radically depending on how your atmosphere is composed and distributed (and other factors, but that’s the main one).
The point about the greenhouse effect is that radiation from the ground gets absorbed by the greenhouse gases, thus heating up the atmosphere. The hotter atmosphere radiates more, and some of that radiation goes back to the ground, making the ground hotter than it would be otherwise. The distribution of temperature from ground to space can change even though the amount of energy lost at the top remains about the same.
[[True, however the concentration of CO2 in the atmosphere is inhomogenous, there is much more at the surface and little at the top, where the photons are ultimately lost to space.]]
CO2 is well mixed throughout the troposphere, due to convection.
[[ What we have is a case of non-equlibrium theromdynamics. Space is a sink for photons and the surface is not. Photons that migrate downward encounter more CO2 molecules and those that go upward less. If you do a random walk deffusion type analysis you will note that the overall transfer is from the surface and into space; the sink will always win.
The question is what effect will the CO2 have on heat trapping? Will an increase in CO2 from 280 ppm to 560 ppm have a large effect on the surface temperature. The answer has got to be no, given that the effect will be about a change in the trapping efficency of about 12%. ]]
The answer has not got to be no, since that would contradict everything we know about radiation physics and the greenhouse effect. Doubling CO2 from 280 ppm to 560 ppm would raise the temperature of the ground 1.2 K by itself, or about 3 K with feedbacks.
[[I have shown two calculation which demonstrate that the half-life of 14CO2 and CO2 is between 9 and 12 years.]]
You’ve been told before why this is wrong, and you just keep on repeating it. You’re tracing the half-life of one molecule or one parcel of CO2 and ignoring the fact that parcels are coming into the atmosphere almost as fast as they are coming out. An individual CO2 molecule will spend about 5 years in the air before entering (say) the ocean. But that doesn’t give you the half-life of the CO2 in the atmosphere as a whole, which is closer to 200 years. Your calculation simply does not take into consideration all the relevant factors. It’s like proving that the Earth’s equilibrium temperature has to be 279 K because that’s the temperature a black iron ball at Earth’s distance from the Sun would have. You have to factor in the Earth’s albedo, and for the surface temperature rather than the equilibrium temperature, you have to get more complicated yet. Your calculations are simplistic and therefore wrong.
[[at the moment with 380 ppm CO2 no IR from the major CO2 absorbtion peals from the surface is tranfered above 10 meters. The only heat being trapped is from IR emissions from the atmosphere. If IR from the surface can’t move more than 10 meters up, why should IR generated at 11 meters be able to move down? ]]
Your 10 meter figure is completely bogus. For a concentration of 400 ppm, 99% of surface radiation is absorbed at 18 meters for the 1.9-2.1 micron band, at 82 meters for the 2.6-2.9 micron band, at 625 meters for the 4.1-4.5 micron band, and for the 13-17 micron band, where the CO2 greenhouse effect does most of its work, at 7,800 meters. Want the math?
Re 257
Dear Dr Martyn,
You are of course correct. The greenhouse effect does not work through back radiation in the way described by Gavin in his Learning from a Simple Model. Explaining his model he wrote:
“Point 1: It’s easy to see that the G (and hence Ts) increases from S to 2S as the emissivity goes from 0 (no greenhouse effect) to 1 (maximum greenhouse effect) … ”
In the case of the planet Venus we have an effective temperature of 227K, and nearly a maximum greenhouse effect of 1. Therefore the temperature of Venus should be 2 * 227 = 454 K but in fact it is 750 K. (Actually the maximum model surface temperature = (2 * 227^4)^0.25 = 270 K.) Therefore, as is known, the models do not work for Venus. See for instance:
‘It’s very disturbing that we do not understand the climate on a planet that is so much like the Earth,’ said Professor Fred Taylor, a planetary scientist based at Oxford University and one of the ESA’s chief advisers for the Venus Express mission. ‘It is telling us that we really don’t understand the Earth. We have ended up with a lot of mysteries.’ Venus: the hot spot
The way greenhouse gases warm the surface was described by Jack Barrett in a notorious paper: Barrett, J. (1995a) �The roles of carbon dioxide and water vapour in warming and cooling the Earth�s troposphere� Spectrochimica Acta 51A, pp. 415-417. Citing McIlveen, he pointed out that all the radiation in the greenhouse gas bands is absorbed in the bottom 30 m of the atmosphere. The greenhouse gas molecules are relaxed by collisions with other air molecules so heating the air. Barrett argued that since all the radiation is already being absorbed, increasing the concentration of CO2 will not cause further warming. However, as Hug has pointed out, with higher concentrations of greenhouse gases the radiation will be absorbed closer to the ground and so the air there will be heated more. From the Beer-Lambert Law it can be seen that if all the radiation is absorbed in 30 m with preindustrial levels (280 ppm) of CO2, then when CO2 is doubled the bottom 15 m will receive the same amount of heat. Heinze Hug claimed that this would not cause global warming because the hot air would convect away. But in deserts the hot air does not convect away. The air density has to be reduced by mixing it with water vapour before it convects. A point to note here is that with the current models it is predicted that the surface temperature will increase with the log of the concentration. With the Barrett/Hug scheme the heating is greater since it increases linearly with concentration.
If Barrett and Hug are right, why then is the planet warming? The answer is that it is more complicated than they describe. The absorption decreases exponentially with distance from the surface. Thus the strongest absorption is closest to the surface. The laminar surface layer below 1cm does not convect and it warms the surface by conduction. Here, is where water comes into play on two fronts. First, the warmer surfaces evaporates more water vapour which is also a greenhouse gas. But more importantly in the present climatic regime, the warmer surface leads to a melting of snow and ice. It is CO2 which is directly leading to the melting of the glaciers world wide, and to the sea ice and ice shelves in the polar regions. This leads to the ice albedo feedback which also causes warming.
One final reference to back all this up is that it agrees with what Philipona is reported. See https://www.realclimate.org/index.php/archives/2005/11/busy-week-for-water-vapor/
re 257
Forgive me, but I couldn’t resist.
“If IR from the surface can’t move more than 10 meters up, why should IR generated at 11 meters be able to move down? ”
Air currents.
I’ll shut up now.
Poking around I see someone with the DocMartyn userid actively posting statements about climate in other forums, obviously having a lot of fun keeping the ball in the air, repeating stuff that gets a lot of attention. I don’t see a lot of learning taking place. I’d like to see some.
If any of these others are the same person, consider getting it together in one place and focusing on basic science. Twenty years ‘doing spectroscopy’ means it’s been a long time since school, eh? Science has moved on a bit.
Arithmetic still works the same though, and logic can still be relied on for basics.
If you gain twelve pounds every winter, and lose eleven pounds every summer, are you gaining weight year after year? If not, why not?
Follow-up to post 102 et seq. about NASA’s Michael Griffin’s arrogant comments regarding global warming: http://www.msnbc.msn.com/id/19058588/print/1/displaymode/1098/
His “regrets” are nearly as misguided as his original comments as they show that his comments were not driven by the scientific data but by his politics. The worst possible background for a supposed *scientific* agency administrator.
Barton Paul Levenson (#266) wrote:
Is there a link or offline article? This sounds like interesting stuff – not that I expect to be able to follow it. But I guess there is always hope.
Marcus, Tim and Ray, thanks for the clarification.
PS to #271
I would assume that a large part of this has to do with the density of the gas, the temperature and thus blackbody radiation which is being emitted at a particular level, then the strength and width of the absorbtion bands. Is this correct? If so, I would gather that the altitude will change as the temperatures at given altitudes change as the result of climate change. Either way, I would still be interested in seeing the math.
Re 270. Dan, Mike Griffin really has no business answering a question like that. He is not a climate expert–not even really a scientist. He is an engineer with a job to do, and as such, one of the most blinkered and narrow-focused animals on the planet. I believe Griffin is convinced that without a reinvigorated manned program that NASA will wither on the vine. He may be right–the US public certainly has given no indication of enthusiasm for the science NASA produces. Thinking that he will also have to shepherd through a bunch of climate satellites and that funding may be drawn away to pay for alternate energy programs is just a nuissance to his achieving his goal.
We keep hearing how complicated the climate is, and that is true. Nonetheless the real issues people need to understand are pretty simple:
1)The energy in Earth’s climate is increasing well beyond the limits of “normal variability”
2)That energy has to come from somewhere
3)Looking at the energy drivers of climate
a) Solar variability can’t come close to explaining the changes we see
b) Water vapor (the most important ghg) can’t come close
c) CO2 (the 2nd most important ghg) can explain it easily
d) All the other energy drivers are pretty far down the scale or aren’t changing
That’s pretty much it. If you get into details, then people can get confused. For instance the fact that CO2 does most of its work in the upper troposphere and above where there isn’t much water vapor is a fact most people miss. And a lot of the potential effects of the warming aren’t all that obvious–e.g. the effects it will have on agriculture, infectious disease, availability of water… That is why the biggest resistance seems to come from either people who don’t have a clue or those who, while educated in their narrow discipline, do not have a broad scientific education that would enable them to fully understand details of the subject. Griffin’s sin was answering a question on an area he really doesn’t understand. That was unwise and unfortunate, but it’s probably should not be a firing offense. If it were, we’d all spend a lot of time in the bread line.
Marcus,
Thank you for your post #262. You have a flair for presenting complex concepts in a manner understandable to laypeople such as myself. I have wondered if much of the difficulty in communicating the science behind AGW is the shortage of images/models/metaphors an average non-scientist can comprehend.
So I’ll pass along (somewhat tongue in cheek) a metaphor I’ve used with skeptical friends.
Many people have heard of the Butterfly Effect, the concept that small perturbations to a system can have large effects. As the theory goes, the wings of a butterfly in the Amazon can trigger a hurricane. Okay, now picture a butterfly weighing 7 gigatons, that’s 14,000,000,000,000 pounds. (That’s more than Oprah and Kirstie Alley combined.) We’ll call it Mega-Mothra. That’s the perturbation our annual consumption of fossil fuels is having on our atmosphere. The consequences won’t be negligable, and they won’t be benign. And remember, this is not a one-time oops, every year we’re hatching another Mega-Mothra out of our smokestacks and tailpipes. A lot of things we hold dear and take for granted are in danger from the effect of this butterfly. There are some who want to mitigate AGW through geoengineering, sort of like building a Robo-Godzilla to battle Mega-Mothra. But we know from experience that everytime we try that Tokyo gets flattened. The only low-risk, long-term solution is to sharply cut back on the CO2 we emit by reducing our consumption of of fossil fuels.
I think that’s a simple enough explanation that even a President could understand it.
Re #273
Timothy, greenhouse gases do not emit blackbody radiation. Greenhouse gases emit line radiation at frequencies that match their vibrotational excitation, not their temperature. The line width depends on pressure so the lines become narrower with altitude, but since the pressure altitude relationship depends on gravitation and mass of the air neither of which change, then that has no effect on global warming.
Well, its not the actual equations, but…
There is more in case people are interested. But part of what I find interesting is the fact that re-emitted photons are more likely to be absorbed if they are re-emitted in the direction of space rather than towards the ground.
Tim, the footnotes to this section might help:
http://www.aip.org/history/exhibits/climate/Radmath.htm
“Callendar himself pointed out in 1941 that the way CO2 absorbed radiation was not so simple as every calculation so far had assumed. He assembled measurements, made in the 1930s, which showed that at the low pressures that prevailed in the upper atmosphere, the amount of absorption varied in complex patterns through the infrared spectrum. Nobody was ready to attempt the vast amount of calculation needed to work out effects point by point through the spectrum, since the data were too sketchy to support firm conclusions anyway.(6)
“Solid methods for dealing with radiative transfer through a gas were not worked out until the 1940s. The great astrophysicist Subrahmanyan Chandrasekhar and others, concerned with the way energy moved through the interiors and atmospheres of stars, forged a panoply of exquisitely sophisticated equations and techniques. The problem was so subtle that Chandrasekhar regarded his monumental work as a mere starting-point. It was too subtle and complex for meteorologists.(7)They mostly ignored the astrophysical literature ….
“Most experts stuck by the old objection to the greenhouse theory of climate change â�� in the parts of the spectrum where infrared absorption took place, the CO2 plus the water vapor that were already in the atmosphere sufficed to block all the radiation that could be blocked. Therefore a rise in the level of the gas would not change anything….
“… In the early 1950s precision measurements at low pressure, backed up by lengthy computations, showed that adding more CO2 really would change how the atmosphere absorbed radiation. While the total absorption might not change greatly, the main site of absorption would shift to higher, thinner layers. And as Callendar had explained, shifting the “screen” in the atmosphere higher would mean more radiation going back down to warm the surface.
…
“… Laboratories began to gather good data only in the 1950s, motivated largely by military concerns.(8)
…
“Some later developments are described in the essay on simple models. However, this Website does not cover technical developments in radiation models (nor details of the other increasingly sophisticated components of general circulation models) from the 1980s forward.”
————–
For science as for history:
“Those who cannot remember the past are condemned to repeat it.”
Much of what’s being asserted is what was believed in the 1930s.
Ray, good and reasonable question (228). The difficulty mostly comes from this: us skeptics express what we see is a chink in the armor. The protagonists reply with an answer. But we don’t completely buy the answer, even though it may be well thought out and researched. That makes it annoying and the protagonist eventually goes away shaking his head and mumbling profanities — maybe rightly so! We come across as irrational (though I’ll contend we’re not) primarily because most skeptics (current skeptical climate scientists excepted) do not have the detailed background or research to back up our concerns. I’m not a climate scientist; I haven’t launched my own satellites or got tied into their data repositories; I’ve never conducted radiometric absorption tests with my (non) lab supply of CO2; I’ve never published my concern in a peer-reviewed journal (or anywhere for that matter); etc. It’s understandable that we are looked upon askance.
But I think our questioning is valid. As long as we have a decent scientific mind and background, especially with ample experience with real world realities, have developed a decent crap detector ( avoiding the normal phrase “bull**** detector”, which implies that the person is knowingly giving out bad information, which is not the case here.) — does it pass the sniff test?, and understands the process where well-intentioned people can err. (The downside is, of course, that us well-intentioned somewhat credentialed skeptics might also be dead wrong.) While we don’t have the premier credentials of the people we are questioning, we’re still under no per se obligation to simply roll over.
So we’re a hard and seemingly (but not really) irrational sell. On that basis I can fully understand why you would eventually give up trying. None-the-less, as long as we maintain ourselves on this side of cantankerous trolls and blind deniers, we’re still in the right even if later we’re proved not right (sorry).
Specific concerns that first come to mind are, in the process arena, the excessive reliance on non sequiturs to “prove” the science: “we out number you 100 to 1” (the consensus shot); “we have way more peer reviewed papers than you skeptics”, and the liberal use of ad hominems (though you clearly don’t have a monopoly here, nor is it universal) toward folks that don’t immediately accept your assertions. These taint your credibility. On the science side, e.g. 1) The argument that when the CO2 in the path length within a constrained bandwidth is absorbing near all of the IR radiation (as it is today in some cases), more CO2 will absorb lots of the radiation outside the nominal bandwidth in some kind of fringe process. CO2 (and other gasses) absorb radiation at a specific resonant frequence (within some bandwidth); It doesn’t sound right that it will start absorbing many photons outside of resonance just because the resonant frequencies are “full”. (btw, many have patiently tried to explain it, but I still didn’t buy it, and they got tired of answering and I got tired of questioning.) 2) I have trouble with the accuracy claimed for measurements — GLOBAL average temperature to 0.1 degree accuracy decades ago (though I tend to accept the slight increase over the last century or so) and satellite measurement of ocean heights to 1mm or even less, then translating those measurements into grand sweeping effects.
There are others, but I’ve worn out my welcome.
RE # 270
Thanks for that link, Dan.
As if Griffin was pained by the controversy he stirred up, he went on to blame the media – not himself.
He said:
[â??Doing media interviews is an art. Their goal is usually to generate controversy because it sells interviews and papers, and my goal is usually to avoid controversy,â??]
Putting both feet into his mouth, he was not yet satisfied. He went on to serve himself even more ridicule.
Obviously, Griffin is not goal oriented. Dangerous for NASA.
Put him out to pasture.
JS has it right, it is the constant emission and absorption of the IR that leads to the greenhouse effect. A good way of thinking about it, is that radiation from the surface heads out. Absorption and emission redirect this into 360 degrees. Another way is to think of what would happen if you shot a bunch of balls (photons) into a bunch of pinball machine bumpers (CO2 molecules). The more bumpers the longer it would take for any one ball to reach the drain (space). To keep the number of balls reaching the drain (keeping the total energy radiated to space constant) per second, you would have to shoot more balls (e.g. heat up the surface so that more photons are emitted per second).
Alastair, Barrett was wrong for a lot of reasons, mostly because he did not account for emission from CO2 molecules and had not a clue about radiative and collisional transfer of energy.
#271 Barton, would you expect the lapse rate to increase at 7 or 8 Km? Been seeing some steep lapse rates during the long Arctic night at about that altitude, specially last December, mainly frequent 7 to 8 C/Km and a few 9 C/Km layers were observed in total darkness. For the steadfast contrarians appropriately mulling over AGW theory, these are strong facts, imagine that, high altitude warm air in prolonged darkness (2 months long).
Pinball Wizard Eli Rabett (#281) wrote:
This is pretty good. I had been thinking about an analogy along much the same lines. However, part of the problem with this approach is that you have the same number of pinball/photons entering the system – assuming the only thing you are varying is the carbon dioxide. So basically, the pinball machine has new bumpers added – representing more carbon dioxide molecules – and as such, the pinballs bounce around more and take longer to get to the bottom. At first, fewer pinballs reach bottom – meaning that more energy is entering the system than is leaving the system. However, as the number of pinballs in the pinball machine increases, more and more pinballs will reach bottom at any given more. More pinballs in the system means more photons which means more energy which means a higher temperature. Balance is achieved once the number of pinballs entering the system equals the number leaving, but with more bumpers, there are going to be more pinballs in the machine for such a balance to be achieved.
Incidently, the pinball and bumpeer idea works well for a good approximation of Beer’s Law, too. If a given number of randomly distributed bumpers results in fifty percent of all pinballs hitting at least one bumper, this implies that half of the pinballs didn’t hit a bumper. Double the bumpers and one-quarter of the pinballs will avoid all the bumpers. Double the bumpers again, and the number will be one-eighth. Of course, this works best if you have tiny pinballs, tiny bumpers, a great many of each, and they are all widely-space.
Eli, Barrett did make several errors but he was right on the one point that the radiation is absorbed in the bottom 30 m of the atmosphere. Oke and McIlveen both agree on that, as does Barton see #266. It is only in most of the rest of the atmosphere that LTE exists.
Thank you for the reference to Evans & Putrin that you posted elsewhere. I have read it, but it contains nearly as many errors as Barrett. Barrett did not say Kirchhoff’s Law was wrong, and he explicly agreed in his reply to Prof. Braterman that he accepted the Schwarzschild equation. Moreover, there are two views of Kirchhoff’s Law and Evans & Puckrin (E&P) proved neither.
First, the law is normally stated as good absorbers are good emitters, or as absorption equals emission in thermal equilibrium. This is trivially true for solids since it follows from the conservation of energy. If the temperature of a body is steady, then any radiation in must equal the radiation out. In E&P the radiation in was from the temperature of liquid nitrogen, and they claimed that the output radiation was at the ambient temperature. That is hardly radiation in equals radiation out!
The second form of Kirchhoff’s Law is that blackbodies radiate with a spectrum that is describe by Planck’s function B(). The spectrum emitted by the CFC was line radiation quite unlike that of a blackbody.
But Kirchhoff’s Law only applies in thermal equilibrium which is not the case for air at the surface of the Earth. Its temperature is continually changing as a result of the diurnal solar radiation cycle.
Schwarzschild’s equation was invented by an astrophysicist for the interior of the Sun. It was the another astrophysicist Chandrasekhar who advocated its use with planetary atmospheres, but the meteorologists have managed fine without it, because it does not apply to the surface temperatures with which they are concerned.
HTH,
Cheers, Alastair.
Alastair McDonald (#276) wrote:
Similar to a laser – where the gas or crystal has only one excitation level responsible for absorption and re-emission, as we are dealing with the same wavelength, but different insofar as it is incoherent and isotropic – although I have heard of a stellar atmosphere actually achieving the stimulated emission of radiation before. Unfortunately the story was at the popular level, and I didn’t dig any further.
In the idealized layer model, they find it useful to treat the atmosphere as if it acts as a blackbody in the infrared – absorbing and emitting all wavelengths. Something to be disabused of. Makes for a more interesting world: there would have been real limits to our understanding of the greenhouse effect prior to the discovery of quantum mechanics. In effect, this is an intersection between quantum mechanics and thermodynamics.
Dear Barton Paul Levenson, Re 266
“Your 10 meter figure is completely bogus. For a concentration of 400 ppm, 99% of surface radiation is absorbed at 18 meters for the 1.9-2.1 micron band”
Whats 10 meters between friends, I was guesstimating the extinction from a google image; however the science reamins the same
“at 82 meters for the 2.6-2.9 micron band, at 625 meters for the 4.1-4.5 micron band, and for the 13-17 micron band, where the CO2 greenhouse effect does most of its work, at 7,800 meters. Want the math?”
No need for the math, I didn’t realise you were going to include the wings. But none the less you believe that incresing CO2 by a factor of two, hence changing the transmission by 12% will heat the world by 2- 5 degrees.
————————————————————————
Tim said in #277
“If an IR photon is emitted lower in the atmosphere, its probability to be (re-) absorbed at a higher elevation is high: Because of the higher elevationâ??s lower temperature, a higher amount of molecules is in an absorbing, lower vibrational state. The opposite is true for atmospheric inversions, leading to a further cooling of the atmosphere already at lower temperatures (example: greenhouse gases generally lead to tropospheric heating due to its negative lapse rate, and stratospheric cooling due to its positive lapse rate).”
I have heard this argument before, and I found it very unconvincing. The logic why this heat transfer method will not work is that it ignore the temperature effect on both absorbtion and emission line broardening.
Now the CO2 molecule has an a complex absorbtion and emmission spectrum, however we can simplyify matters by pretending that the major peak is a gaussian. At high temperatures this peak has a higher width and lower extinction at its peak, compared with at low temperature. So although it is true that the cold gas, with sharpe absorbance peaks, is ready to absorb IR, it has a smaller bandwidth than the IR emissions of hot gas.
It is simple to model, take two gaussians, each with the same area under the curve, but with one 30% or more width. Take one away from the other. You can see that the hot emissions are going to bypass much of the cold gas.
Can I prove it? No. Nor should I have to, this should be done in a lab by people who work in the area. Expirementally is easy, almost all the assumptions made by people who model atmospheric heat transfer are testable, with simple physical expirements, they do not get done.
——————————————————————–
Hank #278
“And as Callendar had explained, shifting the “screen” in the atmosphere higher would mean more radiation going back down to warm the surface.
…”
If true then another way to describe it would mean that the Earths “true surface”, where the temperature matchs 250.22 K you get from appling Stefan-Boltzmann law and 222.3 watts/meter2, has just moved vertically by 500 feet (or 1.2 degrees). Why does the heat have to reach the ground?
——————————————————————
To Eli Rabett #28
“Another way is to think of what would happen if you shot a bunch of balls (photons) into a bunch of pinball machine bumpers (CO2 molecules). The more bumpers the longer it would take for any one ball to reach the drain (space). To keep the number of balls reaching the drain (keeping the total energy radiated to space constant) per second, you would have to shoot more balls (e.g. heat up the surface so that more photons are emitted per second)”
Which assumes that temperature is the only variable, changes in volume or pressure appear to be not allowed. Strange that in the upper atmosphere that an expansion is not allowed, given that it would increase the surface of the radiator.
———————————————————-
Alastair #285.
I can’t hep thinking that people are missing the point of what temperature and heat actually are. You are absolutely correct that the day/night cycle is key. The CO2 arguments outline in this tread would only make sense if we were to observe changes in the rate at which the South pole cools and warms, during its day/nigh cycle. During the whole time that we have been making measurements, there has been no change in the RATE at which cooling and heating occurs. However, during the same time CO2 has increased by 30%. The same arguments for IR absorbtion/emission should be more valid here than they are anywhere, as:
1) The Pole is a desert so there is no need to factor in water vapor changes.
2) There is no conversion of water to ice and vise versa, simplifing everything.
The final points for tonight are these:-
A) If CO2 has a half-life in the atmosphere of greater than 100 years, why and how did 14CO2 in the atmosphere have a decay of 10 years or so?
Did the 14C pixies take it. How can a susbset of total atmospheric CO2 have a decay rate of 10 year-1, if the bulk CO2 has a decay rate of >100 years -1. If you can’t answer this, then all your models are incorrect. No if, no buts, wrong and proven wrong. The 100+ half-life before sequestration does not match the 14CO2 half-life before sequestration. Why and How.
During the day and night cycle, it is both hotter and colder in the desert (Albedo 0.24) than it is in Grassland (0.19 ) or the forest (0.13). Doesn’t this give you some sort of clue to what is pumping out heat during the night and absorbing it during the day?
If the problem of run away green house gases is going to be caused by a decrease in ice reflecting away sunlight, should we be making deserts by bombarding plants with agent orange?
> the meteorologists have managed fine without it, because
> it does not apply to the surface temperatures …
But it does apply at the top of the atmosphere. Don’t forget that.
Rod, do look at the quote I posted pointing to Spencer Weart’s History; first link under Science, right side of the page.
Re #263:
Barton,
Thanks for the explanation. I understood that what you described was happening. It took the later “pinball” analogies for me to get the parts I was missing.
Perhaps a review of Learning from a simple model is in order?
If you look at the first figure you will see a dotted line representing the greenhouse gas layer. As the article notes, it’s a simplification because there are really many layers in an atmospheric model (think of the atmosphere as a stack of transparent pancakes for the purposes of looking at radiative fluxes).
An overview of the historical development is at http://www.aip.org/history/climate/Radmath.htm – and if you want more gory technical details, simply type in “radiative transfer model” in quotes in Google. Here’s one for Jupiter!
Where does this leave the non-atmospheric physicist/modeler? One thing that can be done is to compare the output of these highly complicated models to observations – and the new IPCC report addresses this in a lot of detail. Here’s the link to Chapter 8: http://ipcc-wg1.ucar.edu/wg1/Report/AR4WG1_Pub_Ch08.pdf (6.4 MB pdf file).
The section of interest appears to be 8.3.1.1.2 “The balance of radiation at the top of the atmosphere”.
Let’s start with reflection of incident sunlight:
At most latitudes, the difference between the multi-model mean zonally averaged outgoing SW radiation and observations is in the annual mean less than 6 W m-2 (i.e., an error of about 6%; see Supplementary Material, Figure S8.5). Given that clouds are responsible for about half the outgoing SW radiation, these errors are not surprising, for it is known that cloud processes are among the most difficult to simulate with models
Now, for the infrared �greenhouse� emissions:
At the TOA, the net SW radiation is everywhere partially compensated by outgoing LW radiation (i.e., infrared emissions) emanating from the surface and the atmosphere. Globally and annually averaged, this compensation is nearly exact. The pattern of LW radiation emitted by earth to space depends most critically on atmospheric temperature, humidity, clouds and surface temperature. With a few exceptions, the models can simulate the observed zonal mean of the annual mean outgoing LW within 10 W m-2 (an error of around 5%; see Supplementary Material, Figure S8.7).
However, we live in a fluid environment, so transport processes matter:
For a climate in equilibrium, any local annual mean imbalance in the net TOA radiative flux (SW plus LW) must be balanced by a vertically integrated net horizontal divergence of energy carried by the ocean and atmosphere. The fact that the TOA SW and LW fluxes are well simulated implies that the models must also be properly accounting for poleward transport of total energy by the atmosphere and ocean. This proves to be the case, with most models correctly simulating poleward energy transport within about 10%.
There’s more in there about seasonal variations, and ensemble model runs vs. individual model runs, but that’s the general idea. If you compare the very complex models to the observations, they do match well – and the models are not ‘fit’ to the observations, but rather are based on solid physical principles.
Free hint for skeptics: if you want to attack climate models, there are many other areas you might want to take a look at – the cryosphere, the ocean circulation, the vegetation, the overall carbon cycle… although these are areas where observations seem to indicate that the models are underestimating, not overestimating, the climate response… so perhaps that’s a bad idea.
“At high temperatures this peak has a higher width and lower extinction at its peak, compared with at low temperature. So although it is true that the cold gas, with sharpe absorbance peaks, is ready to absorb IR, it has a smaller bandwidth than the IR emissions of hot gas.
It is simple to model, take two gaussians, each with the same area under the curve, but with one 30% or more width. Take one away from the other. You can see that the hot emissions are going to bypass much of the cold gas.
Can I prove it? No. Nor should I have to, this should be done in a lab by people who work in the area. Expirementally is easy, almost all the assumptions made by people who model atmospheric heat transfer are testable, with simple physical expirements, they do not get done.”
Why yes, that is well known and observed and one of the ways that SOME OF THE emission from CO2 molecules low down escapes. Note that the absorption per molecule at line center is HIGHER for colder molecules. Gilbert Plass was looking at these issues in the 1950s. This is built into all of the radiation transfer codes. Your ignorance is your problem.
Alastair, a molecular in a thermal bath obeys Kirchhoff’s law. The difference is that the absorbtivity/emission is a strong function of wavelength.
“If true then another way to describe it would mean that the Earths “true surface”, where the temperature matchs 250.22 K you get from appling Stefan-Boltzmann law and 222.3 watts/meter2, has just moved vertically by 500 feet (or 1.2 degrees). Why does the heat have to reach the ground?”
Which heat? Energy moves from the surface where the visible sun light strikes the ground and heats it, to the level at which it is emitted to space as IR. The higher the greenhouse gas concentrations, the slower the rate at which energy reaches the altitude where it is emitted. The surface has to warm in order that sufficient energy can reach the layer from which the energy is emitted (it is a few km, not 500 m)
One of the effects of global warming IS to raise the level at which the earth radiates. Of course, the density is also lower so the increase in emission rate is not as large as you would think. This point was made in John Houghton’s reply to Barrett. Y
FurryCatHerder (#289) wrote:
I took a guess that Gavin’s “simple model” could be understood in terms of a series of transfers or “steps” – and modeled it with a static spreadsheet with rows for the steps and columns for each quantity. It worked – giving me his results. But… the pinballs help me, too. Then again, one could use a bath tub analogy – I have heard that before. But I think I prefer the pinballs or marbles.
Maybe it has something to do with having been born after 1905. Nearly anyone in the modern world will tend to think of light as consisting of photons – and when you have photons, you want to know where each one is at.
The bath tub works just fine if you are thinking of it in terms of energy – as if it were water. Turn on the faucet, water pours in, then eventually pours out at the same rate that it enters the tub. Make the tub a little deeper, water will continue to pour in from the faucet, eventually reaching the rim and then pour out at the same rate that it enters the tub – but the tub has more water in it. The increased carbon dioxide consists of the higher walls.
I suppose it is easier to think of water as something continuous but light as something discrete, even though water is made of molecules and photons aren’t strictly particles – but then neither are electrons – since an electron can pass through two slits in the same screen and interfere with itself on the other side. Then again, the tub is missing something: it doesn’t get at the effect of doubling the CO2 like the pinball/bumper analogy does. The tub of water works qualitatively, but the pinballs works both qualitatively and quantitatively. Additionally, the absorbtion and re-emission can be thought of as the before and after of a collision – with pinballs.
Come to think of it, the pinballs are better from a number of different angles – and more than I listed here.
Re #291 Eli,
It is true that the altitude at which carbon dioxide radiates to space is elevated by an increase in its concentration, but this does not affect the amount of radiation. Looking from space you would see a solid wall of CO2 molecules radiating, because all the other air molecules are transparent. Thus the density of the air is irrelevant, as is the height at which this wall appears to be!
If you inspect a spectrum of OLR taken from space you can see that the CO2 band is flat. Ths is because the radiation is coming from the edge of a line emitted at the base of the atmosphere and is not absorbed by molecules above it with narrower lines of absorption and emission.
One mistake that is made it to think that radiative balance to space is achieved by a variation of outgoing longwave radiation. In fact the balance is mainly achieved by clouds altering the incoming solar radiation.
On Earth the clouds are formed from water, on Venus from sulphur dioxide, and on Mars from dust.
Houghton and Shine did not show that Barrett was wrong. Houghton mistook what Barrett wrote (perhaps understandably :-)) and Shine conceded that the models use Planck’s function for blackbody radiation to calculate line emissions. They then both restated the current thinking, but that did not answer Barrett’s criticism of it.
Saying that the surface temperature is set high in the troposphere does not make it true. In fact, that idea is based on the assertion that the troposphere is tightly coupled. The troposphere is rhe region of overturning circulation, which has an inversion above the Boundary Layer. Asserting that the troposphere is tightly coupled does not make that true either!
Re #292 “Alastair, a molecular in a thermal bath obeys Kirchhoff’s law. The difference is that the absorbtivity/emission is a strong function of wavelength.”
Eli, only if the bath is isotropic. If it is being heated (or cooled) by radiation from the Earth’s surface below, it is not isotropic. Treating the atmosphere as an isotropic thermal bath is a simplification too far. It may be true for stellar interiors but it is totally inappropriate for a planetary atmosphere especially one which contains a condensing greenhouse gas.
#295
“Why yes, that is well known and observed and one of the ways that SOME OF THE emission from CO2 molecules low down escapes. Note that the absorption per molecule at line center is HIGHER for colder molecules. Gilbert Plass was looking at these issues in the 1950s. This is built into all of the radiation transfer codes. Your ignorance is your problem.”
I know the extinction coefficient at peak is higher for cold molecules than for hot ones, I even stated it, but what happens at either side of the absorbance peak?
Take two gaussians representing emission and absorbance, give the cold absorption peak half the line width of the hot emission peak. Take one away from the other and you find that 32% of the emission gets through, even though 100% is absorbed at the peak. Its not complicated.
[[The way greenhouse gases warm the surface was described by Jack Barrett in a notorious paper: Barrett, J. (1995a) �The roles of carbon dioxide and water vapour in warming and cooling the Earth�s troposphere� Spectrochimica Acta 51A, pp. 415-417. Citing McIlveen, he pointed out that all the radiation in the greenhouse gas bands is absorbed in the bottom 30 m of the atmosphere. The greenhouse gas molecules are relaxed by collisions with other air molecules so heating the air. Barrett argued that since all the radiation is already being absorbed, increasing the concentration of CO2 will not cause further warming.]]
Barrett is a pseudoscientist. As I show above, the 30 meter figure is bogus. And increasing the concentration of CO2 would cause further warming even if the 30 meter figure were legitimate, because it’s not just absorption of surface IR that matters, it’s absorption among and between all levels.
Re #287
Dr Martyn,
In theory the South Pole would be a good place to test my theory, but its unique features mean that the climate there does not behave in the same manner as the rest of the planet. The South Pole is the only pole that is situated at high altitude at the centre of a continent. (The other pole is at the centre of a small ocean.) Being polar it only has one day and one night during the year, and so it is not suitable for testing normal diurnal behaviour. As a result of its position, its weather is dominated by a polar vortex which brings stratospheric air to the surface. The stratosphere has been cooled by the destruction of the ozone layer as the result of the emission of CFCs. Thus any warming that might be recorded there due to higher CO2 concentration will be offset by the loss of the ozone layer.
Although most people like to think that global dimming was caused by pollution, it was first recorded in Australia far from the Asian Brown Cloud. Investigations in Israel found that it was related to clouds forming earlier in the day, and this would fit with CO2 accelerating the evaporation of surface water, ie an increase in rate of the diurnal cycle.
You are correct that there is a mystery with the fate of C14, but it does not have a bearing on the residence time of CO2. CO2 is cycled through the atmosphere in a period of about 7.5 years. See: http://www.whrc.org/carbon/index.htm Where it all goes to is not known
http://www.whrc.org/carbon/missingc.htm but much of it gets transported to the deep ocean by the thermohaline circulation. That is replaced by CO2 that has remained in the deep ocean for around 1000 years, which is depleted in C14. But since the CO2 which is removed equals the amount escaping from the ocean, then the increase in CO2 concentration due to the burning of fossil fuels will remain, although that CO2 enriched in C13 will also enter the deep ocean.
I did not follow your final remarks, but turning the world into a desert, while it might restore the planetary albedo and lead to a cooler climate does have a major disadvantage. All animal life depends on plants as food, either directly for herbivores, or indirectly for carnivores. They eat herbivores. However, one solution to a reduction in albedo due to a loss of ice would be to increase the albedo of deserts.
HTH,
Cheers, Alastair.