A couple of months ago, we discussed a short paper by Matthews and Weaver on the ‘climate change commitment’ – how much change are we going to see purely because of previous emissions. In my write up, I contrasted the results in M&W (assuming zero CO2 emissions from now on) with a constant concentration scenario (roughly equivalent to an immediate cut of 70% in CO2 emissions), however, as a few people pointed out in the comments, this exclusive focus on CO2 is a little artificial.
I have elsewhere been a big advocate of paying attention to the multi-faceted nature of the anthropogenic emissions (including aerosols and radiatively and chemically active short-lived species), both because that gives a more useful assessment of what it is that we are doing that drives climate change, and also because it is vital information for judging the effectiveness of any proposed policy for a suite of public issues (climate, air pollution, public health etc.). Thus, I shouldn’t have neglected to include these other factors in discussions of the climate change commitment.
Luckily, some estimates do exist in the literature of what happens if we ceased all human emissions of climatically important factors. One such estimate is from Hare and Meinshausen (2006), whose results are illustrated here:
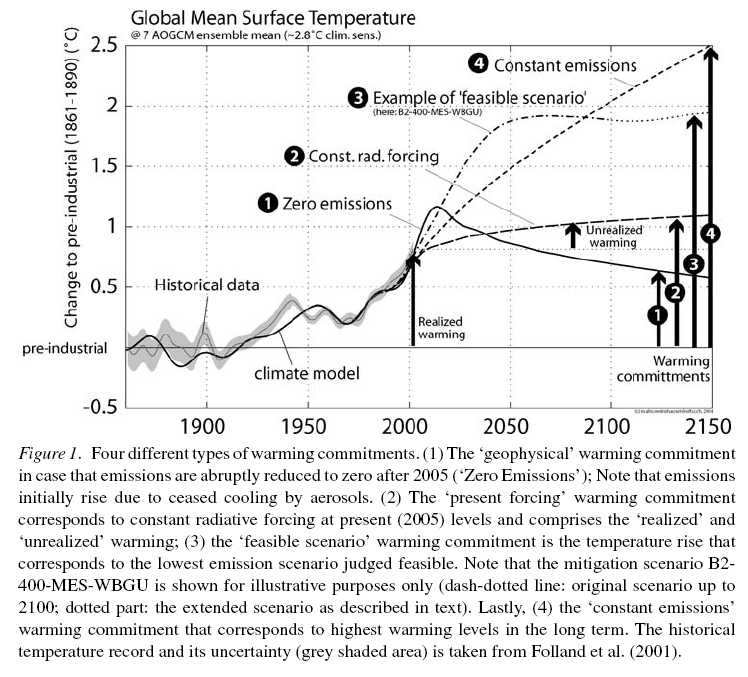
The curve (1) is the result for zero emissions of all of the anthropogenic inputs (in this case, CO2, CH4, N2O, CFCs, SO2, CO, VOCs and NOx). The conclusion is that, in the absence of any human emissions, the expectation would be for quite a sharp warming with elevated temperatures lasting almost until 2050. The reason is that the reflective aerosols (sulphates) decrease in abundance very quickly and so their cooling effect is removed faster than the warming impact of the well-mixed GHGs disappears.
This calculation is done with a somewhat simplified model, and so it might be a little different with a more state-of-the-art ESM (for instance, including more aerosol species like black carbon and a more complete interaction between the chemistry and aerosol species), but the basic result is likely to be robust.
Obviously, this is not a realistic scenario for anything that could really happen, but it does illustrate a couple of points that are relevant for policy. Firstly, the full emissions profile of any particular activity or sector needs to be considered – exclusively focusing on CO2 might give a misleading picture of the climate impact. Secondly, timescales are important. The shorter the time horizon, the larger the impact of short-lived species (aerosols, ozone, etc.). However, the short-lived species provide both warming and cooling effects and the balance between them will vary depending on the activity. Good initial targets for policy measures to reduce emissions might therefore be those where both the short and long-lived components increase warming.
Phil Scadden #547
I have read material on this web site. Here is one I found. It seems to show CO2 responsible for <10% of the downwelling long wave radiation. I have also read that the total amount 100% CO2 could absorb from the entire IR spectrum (at the normal surface pressure and temp) is around 8% as it only will absorb in a few bands of the entire spectrum. This page (based upon measurements) could indicate that all the energy IR radiation CO2 can absorb is being absrobed and then what can return to Earth (via reemission, or being a radiator itself).
Frame 10 has the contribution of CO2 to Downwelling Longwave Radiation.
John E. Pearson
You post: “: Norman, your comments indicate that do not you understand the basic science at all. I would suggest you learn the basic science that you are hoping to disprove. As far as your imaginary experiments not being “a model” how do you figure that they aren’t? You’d like to take the results of laboratory experiments and draw conclusions about planetary atmospheres. That is modeling.”
I am not here to disprove basic science (from all I have read it is not that basic, fairly complex, the underlying science is basic but when you add all the elements together for climate studies it gets vastly complex).
I think there is enough empirial proof to demonstrate a “Greenhouse effect” is a reality. You can measure downwelling Longwave radiation. I did some calcualtions and am just having a hard time grasping how more CO2 will increase, by any significance, the downwelling IR that would keep the Earth warmer than if not happening. My calculations were indicating CO2 is a very IR absorber at its primary frequencies.
I can also understand that IR will behave somewhat different than visible light. I work with a spectrometer on a daily basis. We measure quantities down in the ppb range and some in the PPM range. We use a 50mm sample cell. If the concentration goes above about 50 PPM the source light is completely absorbed and you can’t get a reading without some dilution. So in the world of my reality, it does not take much to absorb all the radiation. I can see the sample material will not gain enough energy to emit any visible light. That is where the difference is with IR, so I can see some of the points. But it would seem that at a point there is too little CO2 in the thin atmosphere above to absorb enough IR to warm that area to become an emission source to produce Downwelling Longwave radiation. That is the parts of the nonsaturation hypothesis that I am working on to understand or reject. But it will take me a bit of time to work on them. I posted because a lot of intelligent people are on this site and can answer questions or may know of source data to help me research this point.
This is the source material I was using to calculate what happens to transmission of IR with increased CO2.
Hitran data for CO2 used in transmission equation.
Equation 7.224 is what I used with the Hitran data.
Nitrogen is 78.09% atmosphere (dry conditions)
Oxygen is 20.95% and CO2 is 0.038%
To recap: I (energy transmitted through medium) = Io (intensity of intitial radiation) e (natural log…Ln) ^ (to power) -S (line strength in cm-1/molecule cm-2) x g(v-vo) x N (molecules/cm^3) x l (path length in cm)
Scientists have calculated that there are about 2.7 x 10^19 molecules/cubic centimeter
Carbon Dioxide would be 1.03 x 10^16 molecules/cm^3
Three times that amount (almost 1200 PPM) 3.09×10^16 molecules/cm^3
I am not sure how to figure Line shape in the equation “g(v-vo)”. I used unity. The Hitran line looked fairly sharp and since it is the same material in both calculations if should not effect the differences.
With the Hitran line strength of 3.5 x 10^-18
400 PPM CO2 1 cm pathlength would transmit 96.5% of the IR radiation
1200 PPM CO2 (same pathlength) would transmit 89.7%
10 cm pathlength at the same concentrations:
400 PPM CO2: 69.7% transmission of IR
1200 PPM CO2: 33.9%
100 cm pathlength (1 meter)
400 PPM CO2: 2.7% radiation will get past this
1200 PPM CO2: 0.002%
10 Meters (1000 cm)
400 PPM CO2: 0 (so small it is insignificant many places below 0)
1200 PPM CO2: also 0
I understand the argument that CO2 will warm by absorbing the Ground based IR and then start emission. But is will be a lower intensity than the ground since CO2 is only absrobing a fraction of the total IR emitted by the ground. So it will emit a lesser amount of IR to the next 10 meter region which will extinguish at 10 meters for both concentrations. That is why I am not getting the warming part of it. Both concentrations will absorb all the IR at their resonant frequency within 10 meters. Even with the idea that the layers above will warm and absorb and emit I can’t see a difference in the effect. Both concentrations should do the same thing.
If you have 400 PPM or 1200 they will both absorb and reemit the same amount of IR all the way up the atmosphere until it leaves for the vacuum of space.
BPL @ 548:
If the PTO would just publish faster …
No, pumped hydro isn’t something you made up, any more than compressed air storage, molten salt, large scale battery banks, or anything else of the sort. However, conservation of angular moment =is= your friend, load-following generation, demand-response, DC interties — these things already exist, and they do a pretty good job of making all those secret magic technologies (what, you gonna just conjure up a hydropower reservoir wherever you need some pumped hydro? Salt domes on demand? The “Genesis Effect” thingy from Star Trek: The Wrath of Khan?) pretty much pointless.
We have 9,000MW of wind installed here in Texas. The other day I was driving back from the Hill Country (client-turned-friend has wind =and= solar!) and saw a pair of blades — giant turbine blades — heading up US87. I see them on IH-35 all the time. What I don’t see is massive aqueducts being built in the Highland Lakes region to turn the LCRA hydropower back around. And yet, I keep on seeing turbine blades heading off somewhere. No flatbeds loaded up with batteries, or salt domes being converted for compressed air storage. And yet those turbines keep on turning …
RL :Gilles says “Again, all absorbed photons are reemitted,…”
BZZZZZZZZZ!!!!!
Oh dear, Gilles, but thank you for playing. See the problem with this answer is that the absorption takes place mainly in portions of the atmosphere that are cooler than the regions of emission, so rather than emitting another photon, most of the excited CO2 relaxes collisionally. It’s called the greenhouse effect. Look it up.”
All process have a reverse process, so collisions can also excite vibrational levels that can also desexcite radiatively. The combination of all that gives a steady photon density that matches the Planck function at LTE. At the strict equilibrium , there is no net creation or absorption of photons – but of course the temperature gradient must vanish at the thermal equilibrium. What you says is correct, but for an optically thick medium , the photon comes from a close region , with a small temperature difference. So what you describe is exactly a heat transfer by diffusion of radiation, but as the difference is small, there is almost as many photons emitted than absorbed, the small difference reproducing exactly the variation of Planck function.
This is only part of the GH effect : the other part is that the atmosphere is transparent in the main part of the incoming radiation, so diffusive transfer is not adapted here : the ground is basically heated directly by incident radiation. So the whole problem is a complex mixture of transport by direct radiation, diffusive transport, and convection (and of course it is not plane-parallel because of the latitude temperature gradient and Earth sphericity). I wouldn’t dare describe it by a couple of simple equations …
Re 553 Norman – If you are finding that 0 % of IR is transmitted over 10 m with CO2 > 400 ppm, then you aren’t looking at enough of the wavelengths. The finer-scale texture aside, the CO2 band centered near 15 microns is shaped such that, letting OptCO2 = CO2 optical thickness per unit distance per unit CO2 concentration, to a first approximation, log(OptCO2) ~= peak value – B*abs(frequency-peak frequency). this means that a 10-fold increase in CO2 will result in CO2 absorbtion exceeding any set threshold over an additional 2*B frequency interval, provided that the peak has already exceeded that threshold. Now, maybe B is different on one side of the band verses the other, so it would be B1+B2 instead of 2*B … it’s an approximation, anyway, but this is why, once the center of the band is saturated, the forcing becomes logarithmically proportional to CO2 (there are some other details about how that works that I could go over, but that will have to wait). Note this relationship can only be extended to a certain point because CO2 has some other absorption bands that become important at very large concentrations (such as on Venus).
In order for a net flux to occur, there has to be some temperature difference over the distances that photons go from emission to absorption (because they can just as easily go in the opposite direction between in two pairs of points, and be just as easily emitted (relative to blackbody intensities) as absorbed (relative to intensities present), at a given frequency). The distance that photons travel is on the order of a unit of optical thickness; optical thickness per unit distance is proportional to the concentration of a material that provides that optical thickness. The net flux depends on the temperature variation relative to distances measured in terms of optical thickness. A radiative forcing (or feedback) occurs when a change in optical thickness causes a change in net flux. An imbalance in fluxes results in some accumulation or depletion of heat somewhere, which tends to change the temperature distribution so as to reduce the imbalance (Planck response).
The surface and troposphere together tend to respond to net forcing changes at the tropopause, because (generally) convection adjusts to radiative imbalances to keep a particular vertical variation in temperature; thus, forcing at the tropopause results in accumulation or depletion of heat somewhere within the troposphere+surface; this results in a temperature change that eventually tends to restore radiative balance at the tropopause level; convection tends to redistribute changes in heat so as to keep the temperature at the surface and various vertical levels shifting together. Changes in the radiative budget at the surface do have effects, but don’t generally have the same overall effect on surface temperature because of how convection tends to adjust in response.
“The distance that photons travel is on the order of a unit of optical thickness”
in average effect
flxible @ 549:
I didn’t say they don’t exist, but look at the values. And while you’re at it, you might want to look at the total hydropower capacity of, say, the Lower Colorado River Authority. Here’s their web page — http://www.lcra.org/energy/power/renewable_energy/hydroelectric.html Now, compare the capacity of the LCRA to the amount of available wind power. Then tell me where all the magic pumped hydro is going to go.
Sources of power tend to be complimentary in odd ways. For example, if the sun isn’t shining (when it should be), quite often the wind is blowing. And the wide open plains that are great for wind — lousy for hydro of any kind. Mountain regions with abundant rain? Too humid for solar.
You can’t just wave your hands, mention a solution, and claim you’ve solved the problem.
Gilles — 13 June 2010 @ 12:25 PM “Again, all absorbed photons are reemitted, since the line is saturated,…”
Norman — 13 June 2010 @ 8:43 PM “If you have 400 PPM or 1200 they will both absorb and reemit the same amount of IR all the way up the atmosphere until it leaves for the vacuum of space.”
Near the surface (1000 mb), the intramolecular distance is small and the average molecular velocity is high (warm and dense), so the mean time between collisions is small compared to the GHG radiative relaxation times; the energy from a photon absorbed by a CO2 molecule is most often transferred to (N2,02). The collisional processes transfer variable amounts of energy to translational, rotational, and vibrational modes. This (thermal) energy gets transferred to H20 and (back) to CO2, and eventually most gets reradiated isotropically, but not necessarily at the same wavelength or even by the same species. Some even gets transported to other parts of the system by non-radiative processes – cloud evaporation, convection. The same thing happens with photons absorbed by H20 and other GHG’s. The upward radiative component moves energy layer by layer up until some of the photons start escaping to space. The altitude at which this starts depends on the wavelength and the concentration of the particular GHG which is absorbing. The altitude at which CO2 starts to emit to space at a concentration of 1200ppmv is further up the atmospheric column than at 400 ppmv. The amount of energy stored in the vibrational states of CO2 is higher per unit volume at higher ppmv concentrations, so even with the same amount of IR radiation from the ground, the partitioning of energy through the atmosphere is different at different CO2 concentrations even when some absorption bands are saturated. At the top of the troposphere (~250 mb) the amount of CO2 above an emitting molecule is about 25% of that in the total atmospheric column, but the amount of H2O is less than 0.1 %, because the decrease in temperature (lapse rate) has caused the H20 to precipitate out. Without the CO2 above the troposphere keeping it warm enough to have an appreciable amount of water vapor at pressures of 300-400 mb, which contributes to maintaining higher temperatures and water vapor at levels of 400-500 mb, and so on to an average surface temperature of about 15 degrees C and a relative humidity of about 60% at the surface, the earth would be an iceball.
Furrie, here’s a couple of links that may help you.
http://www.youtube.com/user/greenman3610#p/u/3/llIbjC49Fjs
http://www.youtube.com/user/greenman3610#p/a/u/2/WO3V2uXTM6k
One highlight for you: the load characteristics follow wind and solar power more closely and therefore the need for as much relative storage to even out load is reduced.
Now it’s a LOT cheaper for Americans especially (but most of the first world too) to reduce power use and it has zero delay: each watt you don’t use is not used RIGHT THEN. Building new power stations to replace coal and oil plants will reduce CO2 production over the time of the farm and that takes time.
Well said, Brian.
It won’t help Gullible and I will bet you not Normal either, but others willing to learn will have done so.
Norman, are you taking into account the fact that satellites can look at the CO2 absorption band and actually see a finite signal? Are you taking into account that the “lines” actually have profile, with a thick tail that extends well away from the central maximum? Are you taking into account pressure broadening. All of this is at least mentioned it the Saturated Gassy Argument.
Perhaps you would do well to try to understand the physics a bit better before jumping into the calculation.
Furry 554: You can’t just wave your hands, mention a solution, and claim you’ve solved the problem.
BPL: Nobody claimed the problem was solved. Just that it wasn’t a very big problem. As you build the alternative energy sources, besides hooking them into wide-area smart grids (the primary leveling method), you build storage as well. The added cost might be 15% of the capital investment cost–which will still leave wind power cheaper than everything else.
Brian Dodge #555
I read what you say but then I look at data like this and it still would seem more of either water vapor or CO2 would not do much over what is already being done, enough warming to make life possilbe on the Planet and water liquid. Are web pages like this just wrong? It does create a sort of confusion. This is not a AGW denier site, it is an Infrared Astronomy page. Telling why you need to send IR telescopes above the atmosphere.
I can’t see the danger presented by CO2 unless another gas is released that absorbs in the IR spectrum that is a window.
IR spectrum most is already blocked at present conditions.
Norman, maybe if I ask a question it will gel the absorption wings issue for you:
Where does the CO2 Absorption band go to absolutely zero? 0.0000 recurring.
#559 Brian”The energy from a photon absorbed by a CO2 molecule is most often transferred to (N2,02)”
Brian, I mainly agree with what you said. You’re right that if the collisional rate is higher than the deexcitation rate, an individual molecule will more often transfer its energy by collision with another molecule than by reemitting a photon. However, by virtue of microreversibility, it is compensated by another phenomenon: that the same molecule will be much more frequently excited by a collision than by a photon, and the small part of these particles that desexcite by emitting a photon produce almost the same number of photons that the absorbed ones. I agree that my formulation wasn’t quite correct : instead of “all photons are reemitted”, I should have said “There are almost as many emitted photons as absorbed ones.” (although not by the same molecules).
This is just to stress that there can be a misconception of “absorbed” radiation, that would predict, for instance, an exponential absorption of the emerging IR line (exp (-tau) ). This would give a ridiculously low level of emitted radiation, and actually it is not correct. The radiation is thermalized by multiple absorptions and reemissions, and the specific intensity is just locally determined by the temperature at the last diffusion surface – not the overall optical depth of the atmosphere. Do we agree?
“All process have a reverse process,”
So unscramble an egg.
flxible (549),
I’ve just been a learning observer on this topic, but I’m now terribly confused. If I read the EIA stats correctly (am I??) I find pumped storage is 2.0% of total U.S. generating capacity and that actual generation in Feb10 was a negative 0.03%, with zero net generation in Texas, contrary to your statement. If I’m reading it right negative pumped storage generation means it generated less than its designated users required. Can you clarify?
BPL @ 563:
As I said, you can’t just wave your hands, mention some technology, and claim the problem is “solved”. And that includes your newest magical incantation — “wide area smart grids”.
You’re absolutely right that wind will be cheaper than everything else, but this isn’t a SciFi novel where you get to claim some magical solution. Most of the existing “smart grid” proposals don’t work, don’t scale, and are a ploy by electric utilities to maintain control over something they are rapidly losing control over. In the words of Malcolm X, “You’ve been hoodwinked. You’ve been had. You’ve been took. You’ve been led astray, led amok. You’ve been bamboozled.”
When TXU Energy stopped paying me for electricity, I just stopped giving it to them (and I have a patent pending on how I did it …) — and that’s why they are wasting our time with the “smart grid” proposals that are floating around out there.
One of the side effects of the growth in the renewable energy market is the ability of individual entities to provide balancing energy services. Bulk production is easy — just keep planting wind turbines, solar panels, micro-hydro, and so on. Balancing energy is the harder part, and it’s a revenue source, the same as bulk production.
If you watch the videos CFU provided, pay attention towards the end of the first one — paying consumers to consume. What if consumers store that power locally, and use it later? Here they’d be paid (and I’ve seen negative pricing in the ERCOT reports — we already have the same problem as Germany from time to time) to consume, but since they’d use their =own= stored energy, they wouldn’t pay when they discharged that storage later. That’s what “balancing energy” is all about. Taking during periods of abundance, returning during slack times, and doing that 5,184,000 times a day (… in North America). This doesn’t require some kind of “wide-area smart grid” — all the information needed to do this is already present. And it scales. And it works. =Today=. All that’s needed is to remove the strangle-hold that the electric utilities have on people like me and my clients.
CFU @ 560:
I think you’re preaching to the choir. I’ve already consulted on three utility-scale solar projects where one issue was ignoring the conventional wisdom and tuning the project, not to maximize output, but to demand.
There is no need to simply reduce power. What we need is to reduce waste, and to work in harmony with the existing grid. We need, as I mentioned to BPL, to remove the obstacles to smaller scale players providing the services that keep the grid running.
Most of all, we need education, and we need people who can debunk the scams that are cropping up like weeds — “Vehicle-to-Grid” has got to be my favorite of all scams, followed by “wide area smart grids”, “turbines are ugly and noise and kill birds” and “solar panels are unsightly” (when a salesdude from Reliant Energy came knocking, I asked if he noticed the solar panels on the roof and he said ‘No’, and I told him that Reliant was evil because they won’t pay for my electricity. Then I invited him to watch a meter spin backwards).
Texas has 9,000MW of nameplate installed wind power, and we got there without V2G and without some “wide area smart grid”. Colorado, as the second video points out, went to 10% of its portfolio 8 years ahead of schedule — no V2G, no “wide area smart grid”.
Norman, It’s secondary to your main question, but I was curious about one piece from your link in #551. The LW downwelling seems to vary significantly over seasons (p5?). It’s not obvious why this would be (as opposed to SW/solar insolation). Can you explain this? (Or am I reading it wrong?)
“by virtue of microreversibility”
Which applies here, how?
Norman (553), I’m a very interested bystander (in this case) on your topic as I have similar questions in this specific area of the physics. One clarification point that doesn’t answer any of your questions but might give some insight As Ray (IIRC) maybe implied earlier (though I’m not sure if he fully agrees), the energy path is 1) earth surface emits IR blackbody radiation which cools (for the moment) the surface. 2) some of that radiation gets absorbed by CO2; this does not change the temperature of the CO2 gas. 3) this now intramolecular energy might re-emit IR radiation — either upward or downward, but is much more likely to de-exite by transferring the absorbed energy to another molecule through a collision; this transferred energy goes into the collidee’s translation and is where you get atmospheric temperature increase. 4) In either case the CO2 molecule is ready to absorb another IR photon.
I don’t know if this is helpful, but that’s what I intend.
“and the specific intensity is just locally determined by the temperature at the last diffusion surface ”
Shorter and clearer: the intensity is determined by the temperature at the 1 optical depth layer of the atmosphere at that frequency.
Just like here:
http://www.statemaster.com/encyclopedia/Optical-depth
http://www.astro.uu.nl/~rutten/rrweb/rjr-material/masters/rtsa/afy.pdf
Gilles (555, et al), like with Norman I have a strong interest in your topic of discussion; you’re both at least playing in the same ballpark. I have a couple of inputs that might be helpful in your endeavor. One, the absorption of the IR radiation by a CO2 molecule is only slightly affected by Boltzmann distribution, but the transfer from another molecule’s translation energy to CO2 vibration energy is highly affected by Boltzmann distribution and is not as immediately likely since the temperature would indicate a very low likelihood of filling a vibrational energy level.
Two, I don’t think photons emitted must necessarily equal photons absorbed, at least short-term.
Three, it sounds like you are saying the earth’s absorbed incident radiation is all SW (solar). Absorbed downwelling LW (IR) radiation (in watts/m^2) is almost twice that of SW/solar absorption. Sorry if I misread what you said.
“There is no need to simply reduce power. What we need is to reduce waste, and to work in harmony with the existing grid.”
We need to do that so that we reduce power usage.
Potayto, potahto.
“We need, as I mentioned to BPL, to remove the obstacles to smaller scale players providing the services that keep the grid running.”
That isn’t what you seem to be mentioning to BPL.
You seem to be mentioning that he knows nothing about this because he’s a science fiction writer.
CFU @ 575:
Why? Most of my clients get to a point where they start to increase energy consumption. If my motorcycle would just get out of the shop, I’d be more than happy to add another 10 or 20KWh a week to my usage. I run my A/C at the coldest I’ve ever run the A/C in my life. I have giant TVes in every room that makes sense. What I reduced was =waste=. But making much of what I use means I get to increase productive consumption, and I may even increase it beyond what I used previously. What you’re doing is feeding the myth that the future of energy is scarcity. In reality, the future is one of plenty — but “plenty” used in an intelligent and creative manner.
The =point= is that bulk power is =easy=. It’s balancing instantaneous production and consumption that’s hard. Solve that (it’s been solved …) and the “plenty” of cheap wind and solar means =more=, not =less=.
I mention that he’s a science fiction writer so people are aware that what he’s mentioning is science fiction, or regurgitated scams.
Here’s some science fact — Texas has 9,000MW of nameplate capacity wind, and routinely produces in excess of 6,000MW from that wind, often exceeding 25% of demand. Please tell me when the super-secret “wide area smart grid” was installed, along with the rest of what he claimed was needed.
Here’s something that’s being done today that improves the grid — run the A/C longer in the morning (it’s summer) and restart it later in the day when the temperature has risen to where it would have been otherwise. This causes the A/C to operate more efficiently (see the 2nd Law of Thermodynamics) and during lower periods of demand. If it improves overall grid efficiency (which it does), the total operating cost will be lower, even if more energy is consumed (which it may). And this can be done right now, no secret magic “wide area smart grid” required.
“Why? Most of my clients get to a point where they start to increase energy consumption.”
Word Smarter, Not Harder.
http://en.wikipedia.org/wiki/Wilkins_Micawber
Why waste more when you don’t have to?
Because you can?
Well, maybe that’s the problem with much of the neo-capitalist world.
“Please tell me when the super-secret “wide area smart grid” was installed, along with the rest of what he claimed was needed.”
Needed? Why do you bring this lump of shapen hay in front of us?
As to the AC idea: again, why are we using energy that we don’t have to?
How about this:
Insulate.
Then the heat from outside takes a day to get in, where you can merely run a fan to exchange the colder air outside for the too warm one inside.
Best of all, it works even better in winter!
Use less.
It costs less in all measures of cost.
CFU: We need to do that so that we reduce power usage
FCU: Why?
Me: Because consumption comes at a cost–even consumption of renewables. Solar cells wear out, as do wind turbines. More consumption means more manufacturing, more resource depletion, more waste and more pollution. We will be supporting 9-10 billion people by 2050. 9-10 billion people cannot continue to consume ever more resources per person indefinitely.
Rod says “2) some of that radiation gets absorbed by CO2;”
True
Rod: “this does not change the temperature of the CO2 gas. ”
Wrong. Rod, S=dE/dT, right? Therefore dT=dE/S, correct? Now S is finite. dE is nonzero. How do you make dT=0?
Gilles: “an individual molecule will more often transfer its energy by collision with another molecule than by reemitting a photon. However, by virtue of microreversibility, it is compensated by another phenomenon: that the same molecule will be much more frequently excited by a collision than by a photon,…”
Absolute BS! The rate of absorptive excitation is determined by the number of photons and the number of molecules. The rate of collisional excitation is determined by the temperature and pressure of the gas. Notice that these parameters are pretty much independent? Your logic is an epic fail.
[edit]
Norman #564, that much of the IR spectrum is “blocked” for purposes of astronomy doesn’t matter to the greenhouse effect. Astronomers would like the actual photons emitted by stars or other faraway objects to hit their eyes/film/CCDs, and a single opaque layer of atmosphere will ruin those wavelengths for them; it doesn’t matter how many more layers you add. Greenhouse warming, on the other hand, goes on and on long after the photons exiting from the top of the atmosphere have ceased to be the same that were emitted from the ground. What matters is how high and cold is the layer of air from which, on average, the escaping photons are emitted; and the more GHGs you add, the higher that will be, and the more the whole atmosphere will need to warm up to restore radiative equilibrium.
“9-10 billion people cannot continue to consume ever more resources per person indefinitely.”
I would put it as “should not continue to consume”.
Even if we could, we should not, without a greater need.
We aren’t in competition against the Joneses, you know.
http://en.wikipedia.org/wiki/Keeping_up_with_the_Joneses
“The =point= is that bulk power is =easy=. It’s balancing instantaneous production and consumption that’s hard.”
cf your request to see all the load balancing of the larger Texas renewable sources of energy.
How come here it’s easy if it’s bulk, but then it was hard BECAUSE it was bulk?
“Wrong. Rod, S=dE/dT, right? Therefore dT=dE/S, correct? Now S is finite. dE is nonzero. How do you make dT=0?”
I would suggest that the point is that the entire atmosphere doesn’t change much if you dump energy into CO2 alone and that the shift in the residency of the CO2 excitation levels do not change much since these in LTE do not get modified a huge amount from the energy absorbed into the bulk.
Therefore the residency of the CO2 excitation levels remains at a low level and can reabsorb more energy readily.
PS that may not have been what Rod meant to say, but it did seem to follow from it and it IS, broadly speaking, correct.
Gilles — 14 June 2010 @ 8:53 AM “…and the specific intensity is just locally determined by the temperature at the last diffusion surface – not the overall optical depth of the atmosphere. Do we agree?”
Yes, bearing in mind that “surface” is a conceptual approximation that represents an arbitrary dividing line in a continuous process. A photon emitted at the bottom of the atmosphere has a finite but possibly very small probability of escaping to space. If it is small enough, it is indistinguishable from zero and treated as such, even though we know it has some finite value. This isn’t “wrong”, but only “inaccurate”, and we can calculate the likely range of errors.
There is some “surface” which we can define as the height above which a photon emitted up (0 to 90 degrees) has probability equal too some arbitrary number of escaping( 1 percent, 10 percent, 1/e). Practically speaking, we can’t count photons, so we – measure – some quantity (e.g. photocurrent) which is a function of the number of photons, but has some inherent noise and uncertainty – measurement inaccuracy. Modeling individual photons and molecules is not practical either, so the atmosphere is approximated by multiple slabs (rather than “surfaces”).
Ray Landbury #562
Your Point: “Norman, are you taking into account the fact that satellites can look at the CO2 absorption band and actually see a finite signal? Are you taking into account that the “lines” actually have profile, with a thick tail that extends well away from the central maximum? Are you taking into account pressure broadening. All of this is at least mentioned it the Saturated Gassy Argument.
Perhaps you would do well to try to understand the physics a bit better before jumping into the calculation.”
There are complications I was not calculating for as you indicate. I looked around a bit and found a person who has done the math and it shows what I am saying. The smearing of the band at higher concentrations is such a small effect that it would not do what is being reported, but I will agree that the effect is there. I am sending a link to the web page so you can look at the math yourself…he doubles CO2 from current levels and does a calculation on how much more IR can be absrobed at the wings of the band.
Calculation of increased absroption by Doubling CO2 from current levels.
[Response: The Hug paper is nonsense and never appeared in the peer-reviewed literature. The criticisms of that paper at the time are still valid. If you want actual science, instead of internet nonsense, read Myhre et al 1998 on the same topic. – gavin]
Ray (580),
How do you explain varying specific heat for a gas?
When Helium absorbs some 668nm radiation by changing electron energy levels, what is the temperature increase of the Helium? When a bunch of CO2 molecules all absorb 15um photons — 1.325×10^-20 joules per molecule — into their vibration energy, what is the CO2’s temperature increase? How do you handle the temperature of the gas using the basic formula: T = [mv^2](avg) / 3k ?
Ignoring for the moment the practical limitations with using the entropy/energy/temp formula, I thought it was dS = dQ/T, not S = dE/dT per your post (E and Q being the same thing).
Re 566 Gilles – “This is just to stress that there can be a misconception of “absorbed” radiation”
I understand what you mean and I can see that your description is in totality of effect approximately what happens when a material is sufficiently opaque that the photon intensity at any location is almost equal to the blackbody intensity for the temperature at that location. However, using the terminology in that way can easily lead to confusion. When a photon is absorbed, it is absorbed; the energy goes into the non-photon matter; that there can be production of photons that might under some circumstances balance the absorption rate doesn’t change this; it is called emission. In between absorption and emission, energy is ‘shared’ among the local population of molecules and the various forms of energy they may have, via molecular collisions/interactions, maintaning a quasi-LTE (so that the population of molecules that absorb and emit radiation transfer their energy gains and losses to the whole local population, with molecules gaining energy so that they can emit photons at a rate according to the temperature of the whole local population). The photons that are emitted are not the same photons that are absorbed; they are new photons. The net effect can be zero net absorption when the intensity of photons present match the local blackbody value, but the distinction is important. When the photon intensity is not equal to the local blackbody value – that is, when the brightness temperature of the radiation is different from the temperature of the non-photons (for quasi-LTE conditions), there is some net absorption or emission (thus causing a spatial variation in the flux in some direction, and if the spatial variation of flux in one direction is not matched by the spatial variation of flux in the opposite direction, there will be some net flux convergence or divergence along that direction, which is equal to an accumulation or depletion of energy). There are wavelengths for which the brightness temperature can diverge significantly from the local temperature within the atmosphere.
The misconception that the intensity of radiation just drops exponentially to approach zero is based on using Beer’s law when Schwarzchild’s Equation or some extension of that would apply (the former doesn’t include emission along a path; the later does).
…(thus causing a spatial variation in the flux in some direction,…)
Actually, that might not be the best and most general way to explain what happens, but, but it applies in the case of isotropic (or at least 2-fold rotational symmetry)optical properties, which generally applies to atmospheric conditions for LW radiation, at least for randomly oriented gas molecules and spherically-symmetric or randomly-oriented particles).
… no, actually I think that should work even in more general cases …
… In so far as emission and absorption dominate, the radiant intensity’s brightness temperature is, over distance, tending to approach the temperature of the region it passes through (it does this more rapidly over distance when the material is more opaque). For LW radiation in the Earth’s atmosphere, that’s good enough for now, but there can be additional complexity when there is scattering and reflection (the spatial trend in brightness temperature can start to depend on variations between temperature all around the location considered. (My last two comments were made while considering scattering and reflection; and also the oddball case of particles that are absorbing on one side and reflective on the other, but that’s not a particularly common thing so far as I know and wouldn’t matter if they were randomly oriented anyway.)
R.L.”Absolute BS! The rate of absorptive excitation is determined by the number of photons and the number of molecules. The rate of collisional excitation is determined by the temperature and pressure of the gas. Notice that these parameters are pretty much independent?”
of course they are – but IF the rate of collisional deexcitation is much larger that the rate of radiative deexcitation, then it means that the pressure must be high enough (since the latter is pressure independent), and it turns out to yield exactly the same condition for collisional excitation being larger that radiative excitation, if radiation is thermalized. Just because the rate of excitation to deexcitation rate must be equal, both for radiative and collisional processes, for a near LTE (quasi Boltzmann distribution).
“Wrong. Rod, S=dE/dT, right? Therefore dT=dE/S, correct? Now S is finite. dE is nonzero. How do you make dT=0?”
what is S? entropy? then your equation is wrong , it is T = dE/dS, and it has very little to do with the present discussion. This is a transport problem, and it has little to do with a “warming” of the atmosphere by absorbed radiation – as I said the net radiative budget of any volume is ZERO at steady state.Only the global solution including the boundary condition at the ground and at the upper last diffusion surface, gives a higher gradient, and thus a higher ground temperature if you increase the GHG concentration.
Rod re pumped hydro storage
I was just responding to the apparent assertion that the technology was science fiction, because I knew it’s not. From the footnotes: “Negative generation denotes that electric power consumed for plant use exceeds gross generation”
It uses energy to pump to storage, you’re not going to generate that extra in return – as for Texas, I just noted it’s on the chart, meaning [I assume] the capacity is there but not much used.
Rod, when you place a population in inversion, you not only change the temperature, you make it negative! Adding energy changes temperature unless you make the system change follow a very particular path. Temperature has to do with much more than kinetic energy, I promise you.
Re 589 Rod B – the specific heat cv = Du/DT where u is internal energy per unit material; internal energy includes translational kinetic energy but also molecular potential energy and various forms of energy within molecules (vibration, rotation, electronic excitation when that happens, etc.). Molecules with more atoms tend to have higher cv because of the greater number of vibrational and rotational modes. Because these are quantized, they are not ‘available’ at all temperatures, so cv can be lower at lower temperatures. Dq (where q is Q per unit material) = Du + p*Dα, where α is the volume per unit material and α*Dp is work done by expansion (this relationship of course excluding (I think) any non-mechanical work – electrochemical stuff, etc. …)… cp is the specific (or molar, depending on how amount of material is measured) heat at constant pressure; cv is the specific (or molar) heat at constant volume. See http://chriscolose.wordpress.com/2010/05/12/goddards-world/#comment-2415 – but don’t trust my derivation of cp – cv for non-(ideal gases); it disagrees with the wikipedia version. See also http://hyperphysics.phy-astr.gsu.edu/Hbase/heacon.html#heacon
Correction: p*Dα is work done by expansion, not α*Dp
CFU @ 578:
[edit]
My patents pending require NO WIDE AREA SMART GRID!
It was the true brilliance of them. Because the existing smart grid proposals don’t work anyway. We went through a ton of prior art and “does it scale?” was one of the big questions. For a lot of the ideas out there already, “No” is the answer. “What are the common failure modes, and does it survive them?” was another that elicited a lot of “No” answers.
Because I live in Texas where there is a very distinct possibility that there is no source of “colder air outside”. There are also issues of humidity control, which can’t be fixed by pumping all the hot air out and replacing it with cold air.
The other problem — and this is a fun one — is that running appliances indoors produces heat. Oh, and people make a LOT of heat and give off a lot of moisture.
It’s not just that pesky outdoor heat, it’s the indoor heat as well, plus the indoor respiring human beings and our damned soggy breath.
Comment from one person on the “Hug” paper I linked to above.
“Although the radiative calculations used by the IPCC might be basically correct, the modeler must have some view of the atmospheric water distribution and I don’t think anybody can model the complicated connections between evaporation, condensation,convective heat transfer, radiative heat transfer, clouds, water content a.s.o. They may not even know enough about the influence of the composition of the atmosphere on the radiative properties.
What we can learn from this discussion, is that it may be a good idea to perform more laboratory experiments using modern spectroscopic equipment and realistic temperatures for the “black body” source and the temperature, pressure and chemical composition of the absorbing/emitting gas. We have seen too much of computer simulations, probably using garbage input…. ”
Jarl R. Ahlbeck D.Sc.(Chem Eng.)
Research Associate,
Abo Akademi University, Finland
The point he is making is similar to what I am asking. Maybe it is out there but hard to find. Where is some good lab testing for proof of some ot the AGW theories? How much more actual energy does a double of atmospheric CO2 send in downwelling IR. Although some may disagree with me, setting up a lab test to get as close as possible to an atmosphere (like individual sealed containers at various pressures that create a large column to beam the same IR spectrum the Earth gives off…can be varied to match different parts of the Earth at different seasons) is not the same as a computer simulation. Most the posts I am reading in response to my questions are theory and ideas but lack some solid data to back up the idea.